Unleash the power of our Recombinant Human ISCU protein in your signal transduction research, and explore its role in the vital process of iron-sulfur cluster biogenesis. ISCU, the Iron-sulfur cluster assembly enzyme, is a critical component in the assembly and transfer of Fe-S clusters, which serve as essential cofactors for a multitude of cellular processes, including electron transfer and enzyme catalysis.
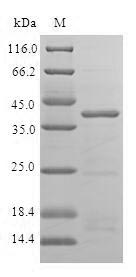
Our Recombinant Human ISCU protein offers a full-length sequence (1-167 amino acids) for optimal functionality and bioactivity. Produced in E. coli, this protein features an N-terminal GST-tag, facilitating efficient purification and detection without affecting the protein's native structure or biological activity. With a purity of greater than 90% as determined by SDS-PAGE, our Recombinant Human ISCU protein guarantees consistent and reliable results, providing a dependable tool for your research endeavors. Choose from liquid or lyophilized powder forms to best suit your experimental needs.