What Is Axonal Regeneration?
An axon also called nerve fiber, resembles a cable extending from the soma along which electrical impulses are conducted away from the cell body to other neurons, muscle cells, or gland cells. It is the basic unit of structure and function during nerve impulse transmission. During development, axons extend and create a well-crafted circuitry that allows for a functional nervous system.
Axon regeneration is a highly orchestrated process involved in multiple cellular events, including but is not limited to injury sensing, axonal trafficking, macromolecule synthesis, cell energy homeostasis, and cytoskeletal organization. It includes three phases: sprouting, elongation, and maturation.
Mechanisms for Axonal Regeneration
The growth of axons starts from the growth cone, which can determine the direction of axon growth and the length of extension by sensing the information of the external environment. In the nervous system, growth-promoting factors and growth-inhibitory factors synergistically regulate axon growth cones.
The prerequisite for axon regeneration after injury is to rebuild a growth cone at the tip. The synthesis of a new growth cone is a multi-processes event, including calcium signaling, restructuring of the cytoskeleton, transport of materials, local translation of messenger RNAs, and the insertion of new membrane and cell surface molecules.
Following the injury to the mature mammalian central nervous system (CNS), lesioned axons fail to regenerate and re-establish functional connections with their original targets, leading to permanent disabilities of neural circuits.
Inhibitors of Axonal Regeneration
Axons in the CNS fail to regrow spontaneously following injury, such as spinal cord injury (SCI). Two primary obstacles to axonal regeneration are adult CNS neurons' diminished intrinsic regenerative capacity and extrinsic growth-inhibitors that hinder axon regrowth. External axon regrowth inhibitors are described below.
Formation of the Glial Scar and Secretion of Related Inhibitory Factors
Inflammatory reactions boost significantly at the damaged area shortly after SCI, leading to cavitation that enlarges the initial lesion. And then, reactive astrocytes enclose this injured site to form the glial scar, which is a dense structure mixed up with extracellular matrix and various cell types. Although the glial scar is conducive to controlling inflammation and reducing further damage, it can cause axonal growth cones to collapse, thereby hindering neuron reconstruction and repair.
In addition, reactive astrocytes produce and secrete many chondroitin sulfate proteoglycan (CSPGs) after injury. Other growth-inhibitory factors are also up-regulated, including glial fibrillary acidic protein (GFAP), keratan sulfate proteoglycan (KSPGs), tenascin, and RGM (RGMA, netrins, sema, ephrin, and slits).
CSPGs are currently the most studied and are mainly composed of a protein backbone and glycosaminoglycan (GAG) chains connected by covalent bonds. GAG chains are responsible for the inhibitory properties of CSPGs. The core protein is N- and O-terminally connected to different types and quantities of oligosaccharides or polysaccharides. Recent studies have demonstrated that CSPGs are involved in remyelination deficits as well, further contributing to regeneration repression. CSPGs have also been shown to suppress the process of outgrowth and differentiation of oligodendrocyte progenitor cells in vitro.
Formation of Myelin-derived Inhibitory Factors (MAIFs)
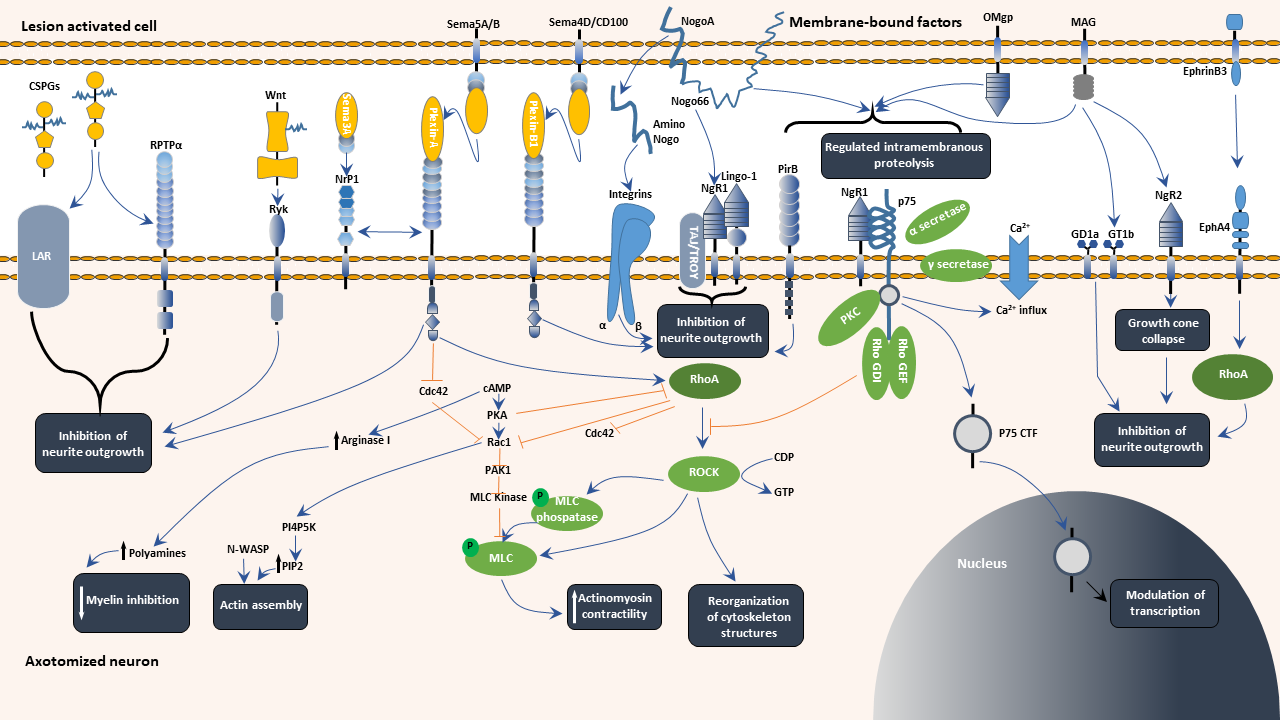
Early studies have shown that CNS myelin sheath contains axonal regeneration inhibitory factors. Myelin-derived inhibitory factors (MAIFs) are highly expressed in the glial scar, inhibiting the differentiation of oligodendrocytes from the injured part to neurons, reducing axon regeneration, thereby affecting the repair of damaged nerves. MAIFs mainly contain myelin-associated glycoprotein (MAG), neurite outgrowth inhibitor protein (NOGO)/Reticulon-4, and oligodendrocyte myelin glycoprotein (OMgp).
NOGO is divided into three isomers: NOGOA, NOGOB, and NOGOC. Among them, NOGOA is the most specific and the only one highly expressed in oligodendrocytes. MAG is another intrinsic molecule in the myelin sheath that can effectively inhibit axon regeneration. It is a transmembrane protein with 5 immunoglobulin-like domains in the extracellular region. In addition to the above two inhibitors, OMgp is also an endogenous inhibitor of CNS myelin upregulation. OMgp has 5 leucine-rich repeat regions (LRR) and an N-terminal flanking region. Although these intrinsic inhibitors have no structural similarities, they have a high affinity for NOGO receptors (NgR). NgR has 8 leucine-rich LRRs and a C-terminal chain at the N-terminal end.
The ephrin B3, ephrin A3, semaphorin 4D, semaphorin 5A, semaphorin 3F, as well as the myelin glycolipid sulfatide were all found to exert strong growth inhibitory effects on various neuronal cells in vitro.
Sina Stern et al. found that neuronal ras homolog gene family member A (RhoA) inhibits axon regeneration by activating myosin II to compress actin and so control microtubule protrusion in response to external inhibitors. Astrocytic RhoA, on the other hand, limits injury-induced astrogliosis via stimulating Yes-activated protein (YAP) signaling in myosin II, which is independent of microtubules. Neuronal RhoA activation hinders axon regeneration, whereas astrocytic RhoA help to regenerate axons, according to cell-type-specific deletion in spinal-cord-injured mice. This finding has indicated that cell-specific RhoA targeting is a promising strategy for the treatment of axon regeneration limitation.



