What Is Leukocyte Transendothelial Migration?
During inflammation or immune surveillance, leukocytes in the blood pass through endothelial cells lining the luminal site of blood vessels and then migrate to the underlying tissue. The process is called leukocyte transendothelial migration (TEM). Leukocyte transendothelial migration occurs across micron-wide gaps in the specific post-capilary venules produced by the transmigrating leukocyte.
The Function of Leukocyte Transendothelial Migration
Leukocyte transendothelial migration is indispensable to initial the innate or adaptive immune response. In other words, leukocyte migration from the blood into tissues is vital for immune surveillance and inflammation. The inflammatory response is the body's defense mechanism against bacterial infection, cell damage, foreign irritants, and other adverse factors.
The Mechanism of Leukocyte Transendothelial Migration
When the inflammatory reaction occurs, endothelial cells attract and direct leukocytes (or other immune cells) to move across the vascular endothelial cell membrane to the inflammatory site, where leukocytes exert their physiological functions.
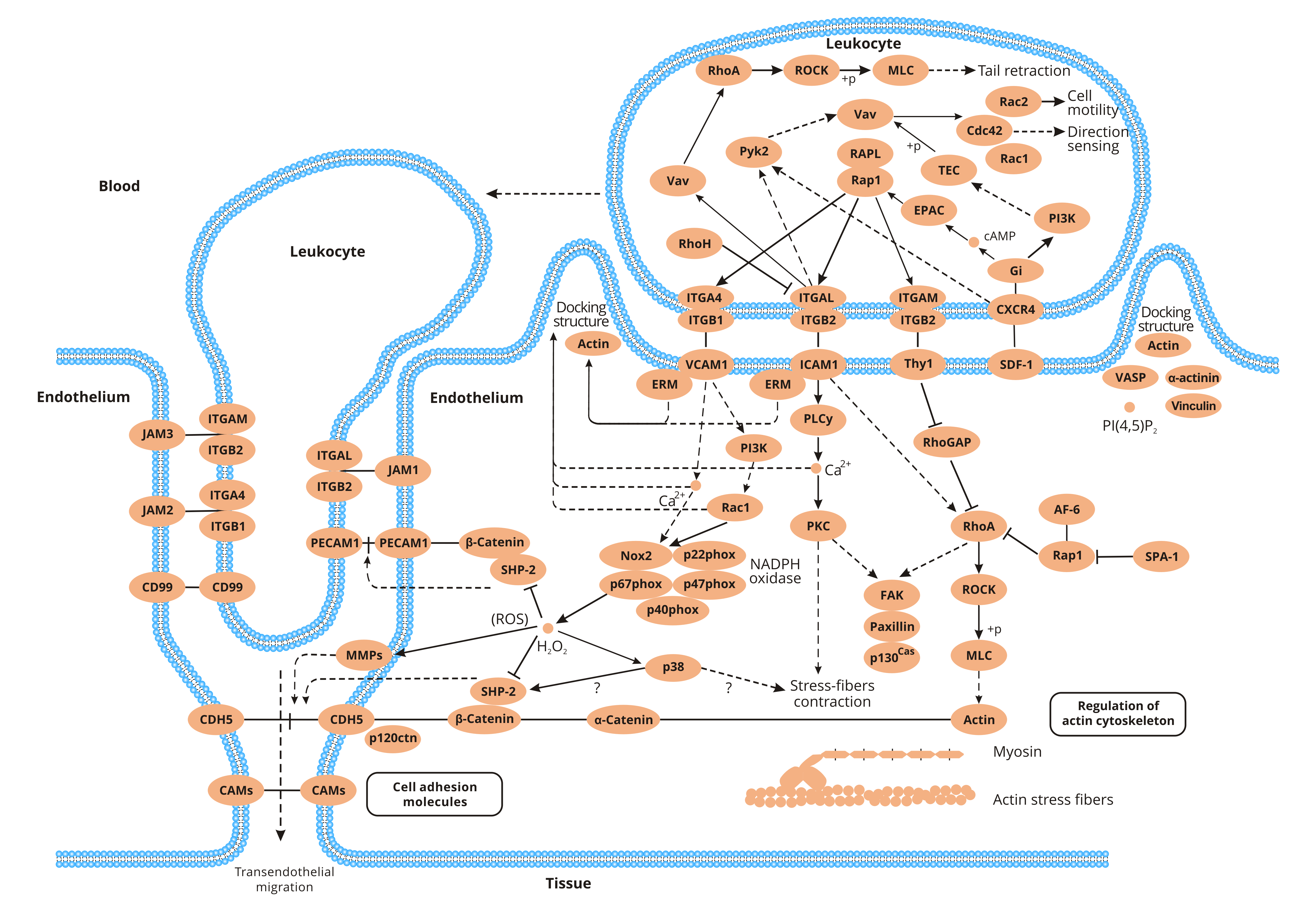
Leukocyte transendothelial migration is a complex and multi-step process. It is the result of interaction between receptors expressed on leukocytes and ligands on endothelial cells. Endothelial cells contain a dense cytoskeletal network, so leukocyte transmigration must overcome these mechanical barriers as leukocytes squeeze their nuclei through endothelial gaps and pores. Recent findings suggest that endothelial cells are not a passive barrier. And upon engagement by transmigrating leukocytes trigger extensive dynamic modifications of the endothelial actin cytoskeleton. The whole process includes the rolling of leukocytes, leukocyte activation & arrest/adhesion, and transendothelial migration.
Various inflammatory factors and adhesion molecules are involved in the process of leukocyte transendothelial migration. They control the morphology and movement of leukocytes and endothelial cells by regulating the dynamic recombination of the cytoskeleton in endothelial cells to realize the leukocyte transendothelial migration.
The rolling of leukocytes
The systemic circulation of leukocytes through the veins slows down or pulls over initiates migration of white blood cells during inflammation. The edge phenomenon refers to the movement of leukocytes in the bloodstream of the venules near the endothelial surface of the vessels.
Selectins mediate leukocyte capture into the vessel wall. L-selectin is structurally expressed on leukocytes, which facilitates the binding of leukocytes in the flowing bloodstream with L- selectin ligands on vascular endothelial cells to catch leukocytes. Leukocytes adhere through L-selectin to cellular adhesion molecules (CAM) or glycoproteins such as CD34 on the endothelial cells. L-selectin induces CXCR4 upregulation from intracellular granules on the leukocyte surface, resulting in increased activation of Rap1. Rap1 induces α4-integrin activation. Adhered leukocytes move forward by leading-edge protrusion and retraction of its tail. The rolling of leukocytes along the endothelia gives them prolonged contact with endothelial cells, allowing them to make effective contact on the surface of endothelial cells.
Leukocyte activation & arrest/adhesion
Leukocytes use mainly β1 integrin (VLA-4/α4β1) and β2 (LFA-1/αLβ2) integrin to firmly adhere to the endothelium. The ligands for these integrins on the endothelium are respectively VCAM-1 (vascular cell adhesion molecule-1) and ICAM-1 (intercellular cell adhesion molecule-1).
The β1/β2 integrin activates leukocytes and allows them to adhere more firmly to the endothelial surface. Activated endothelial cells produce chemokines such as IL-8 and PAF. IL-8 is one of the major endogenous chemokines that activate neutrophils, causing a series of changes between cells that eventually lead to cell migration.
Transendothelial migration
The formation and depolymerization of focal adhesion play an important role in the migration of leukocytes across the endothelium. The small GTPase Rac1 induces ruffles, possibly leading to docking structures and ROS production. ROS suppresses phosphatase activity, which causes increased kinase activity and loss of VE-cadherin- mediated cell-cell junctions. The small GTPase RhoA subsequently induces activation of MLC and cell contraction. All these processes, together with junctional molecules such as PECAM-1, JAM, and CD99, are likely to promote the transmigration of leukocytes across an endothelial monolayer.



