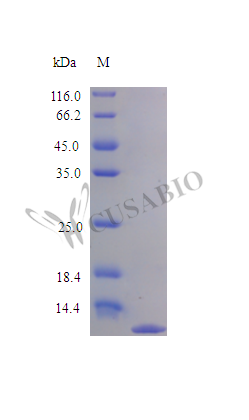
HHV-8 VMI2 functions as a broad-spectrum chemokine antagonist that binds to multiple host chemokine receptors, thereby inhibiting leukocyte migration and facilitating viral immune evasion. This tag-free partial recombinant HHV-8 VMI2 protein (aa 24–93) is produced in E. coli, exceeds >97% purity by SDS-PAGE, and exhibits specific inhibitory activity against monocyte migration with 1.0–10.0 µg/ml of viral MIP-2 inhibiting 25 ng/ml of human MIP-1 alpha—supporting direct use in chemotaxis inhibition assays, viral immunomodulation studies, and HHV-8 pathogenesis research. Endotoxin levels below 1.0 EU/μg as determined by LAL method eliminate LPS-driven artifacts that confound monocyte and innate immune cell functional assays, making this preparation appropriate for in vitro migration studies where contamination-free stimulation is critical. The fully biologically active HHV-8 VMI2, confirmed by comparison to standard, combined with stringent endotoxin control and tag-free sequence, provides a reliable tool for dissecting viral macrophage inflammatory protein interactions and validating antibodies targeting VMI2 in immunological applications.