Human Leukocyte Antigen, A Guard of Human Body Immune System
In 1958, Jean Dausset first discovered that the serum of patients who had been rejected after kidney transplantation and who had received multiple transfusions contained circulating antibodies that could react with the donor's white blood cells. This antibody-targeted antigen was named major histocompatibility complex (MHC) due to its role in graft rejection and tissue compatibility between donor-recipient pairs.
MHC genes are a group of closely linked gene clusters that encode major histocompatibility complexes in all jawed vertebrates. MHC controls mutual recognition between immune cells and regulates immune response. The human MHC-encoded glycoprotein is also called human leukocyte antigen (HLA), which has become an important emerging research field in immunogenetics, immunobiology, and biochemistry by the 1970s.
This article mainly describes the multi-angle characteristics of human leukocyte antigens including genetic, structural, and functional aspects, as well as their related diseases and drug research.
1. Human Leukocyte Antigen (HLA) Complex
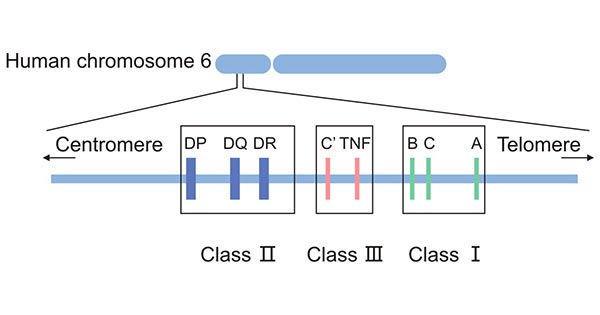
The HLA complex is located on the short arm of chromosome 6 (6p21.3) and contains more than 200 gene loci, spanning about 3600 kilobase DNA [1], with a length of about 4cm [2] and accounting for 1/3000 of the whole human genome. It encodes a family of proteins or makers present on the surface of most cells in the human body. These HLA proteins are used to recognize which cells belong in the body and which do not.
The HLA complex is located in one of the most polymorphic regions of the human genome, with up to thousands of alleles. It is characterized by polymorphism, linkage disequilibrium, co-dominance, and haplotype heritability. It can be divided into four regions from the centromeric side, including D, C ', B, C, and A regions.
2. Human Leukocyte Antigen Function
The HLA molecules specialize in presenting short peptides to T cells and primarily function to induce and regulate immune responses.
HLA is an important molecular marker for identifying human leukocytes. It is also the basis for the immune system to distinguish itself from foreign substances, with very important biological functions.
Additionally, many reports have pointed out that HLA is one of the disease-related genetic markers and is related to the process of some infectious diseases, maternal and infant immune responses, tumors, and other diseases.
The HLA complex is highly polymorphic, and its extreme polymorphism has possibly evolved as a mechanism to deal with all of the different peptides that will appear. The probability of identical HLA phenotypes among unrelated individuals is extremely low, so the HLA complex is considered a specific genetic marker that accompanies individuals throughout their lives.
3. The Classification of HLA
HLA complex consists of dozens of loci, which can be traditionally classified into three categories based on the structure, expression, tissue distribution, and function of their products, including HLA class I, class II, and class III.
Figure 1. The classification of HLA
3.1 HLA Class I
HLA class I proteins are expressed on the surface of all nucleated cells with different levels, while mature red blood cells, nerve cells, and trophoblast cells do not express them. They are highly expressed in peripheral blood leukocytes, lymph nodes, and spleen cells, followed by the liver, skin, aorta, and muscle. Classical HLA class I includes HLA-A, HLA-B, and HLA-C.
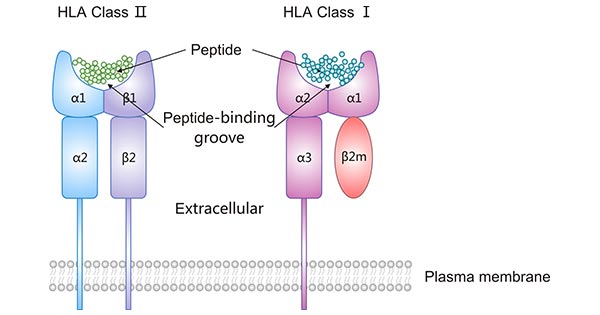
HLA-I antigens contain three a glycosylated amino acid heavy chain, a β-microglobulin (β2m) light chain, and a short self-peptide (8-10 AA). Their heavy chain is composed of a cytoplasmic region, a transmembrane region, and an extracellular region consisting of three immunoglobulin-like domains (α1, α2, α3).
The α1 and α2 domains form the peptide-binding groove, which is the site responsible for the binding pockets for the foreign antigen-derived peptide and subsequent presentation to peptide-specific CTL for scrutiny. The α3 domain has a CTL-expressed CD8-specific binding site [3] and helps boost the killing of virus-infected cells or cancer cells.
β2m plays an essential role in the correct conformation of the peptide-binding groove of the heavy chain and stabilizes the HLA class I antigen-peptide complex on the cell surface. It thus indirectly contributes to the antigen presentation to CTLs' specific T-cell receptors [4-6].
HLA class I molecules normally function to present expired or defective intracellular protein-derived and invasive virus-derived peptides to the T cell receptor (TCR) on CD8+ cytotoxic T cells (CTLs), resulting in the disruption of the cells by immune mechanisms. They also bind to killer inhibitory receptors (KIR) expressed on the surface of NK cells, causing suppression of NK cell activity [7].
3.2 HLA Class II
HLA Class II molecules include HLA-DR, HLA-DP, and HLA-DQ and are especially expressed on immune active cells, including B cells and antigen-presenting cells such as monocytes/macrophages, dendritic cells, as well as activated T cells. They bind to CD4+ helper T cells to present exogenous antigens.
An HLA class II protein consists of α (α1, α2) and β (β1, β2) chains, each of which has two extracellular domains. The α1 and β1 domains assemble into the peptide-binding groove that always carries a self- or non-self-protein-derived peptide coming from outside the cell by endocytosis.
Figure 2. The structure of HLA class I and HLA class II
HLA class II proteins and their bound peptides interact with CD4+ T cells (usually Th cells) and their receptors. The affinity of peptides for binding to HLA class II molecules will affect the allergenicity presented and recognized by TCR.
Table 1 The composition of HLA/MHC Class II
3.3 HLA Class III
Proteins produced by HLA class III genes are immune regulatory molecules, including complementary components C3, C4, and C5, tumor necrosis factor (TNF), lymphotoxin, and heat shock proteins. They are uniformly distributed in the plasma in soluble form and mainly play a role in the inflammatory response.
4. HLA Antigen-processing Pathway
Cells including all nucleated cells (except for mature erythrocytes) and B cells as well antigen-processing cells have two different antigen-processing pathways that present peptides to T cells.
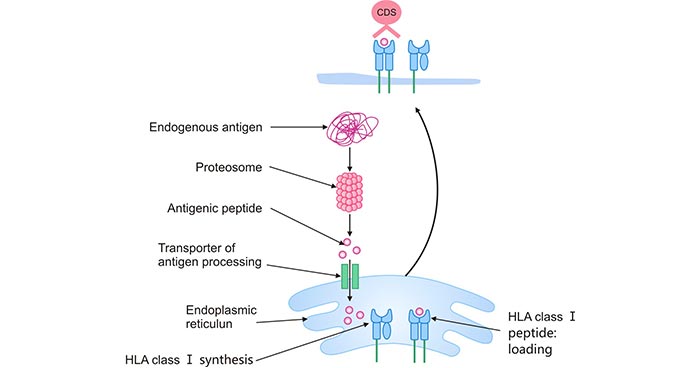
4.1 HLA-I antigen-processing pathway
HLA class I molecules load peptides on the endoplasmic reticulum (ER). These peptides are degraded products of endogenous antigens such as cytosolic and nuclear proteins by proteasomes.
Selected peptides are translocated into the ER via transporters associated with antigen presentation (TAP). These peptides may undergo further trimming by aminopeptidase ERAAP1 and ERAAP2 in the ER to better binding to HLA-I. The HLA-I-peptide complex is exported by the Golgi apparatus and presented on the cell surface of the CD8+ T cells [8][9]. Eventually, the HLA-I-peptide complex binds to TCR-activating CTL which releases perforin and granzyme B (GzmB) to kill the infected cells.
Figure 3. HLA class I antigen-processing and presentation pathwayI
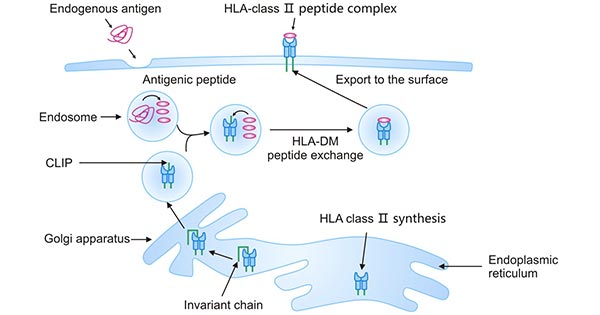
4.2 HLA-II antigen-processing pathway
The HLA class II molecules are synthesized in the ER and form a complex with the invariant chain (Ii) that is bound to its peptide-binding groove. The invariant chain directs HLA-II transport to the Golgi, which is further fused with late endosomes. During endocytosis, the invariant chain is degraded by cathepsins, and its particular amino acid sequence, the class II related invariant chain peptide (CLIP) is retained in the binding groove. HLA-DM subsequently mediates the exchange of CLIP and antigenic peptides, a process known as peptide editing. The HLA-II-peptide complex is transported to the cell surface and presented to CD4+ T cells [10].
Therefore, HLA class I usually presents intracellular peptides to CD8+ Tc cells for scrutiny, while HLA class II detects extracellular peptides and delivers them to the cell surfaces to be scrutinized by CD4+ Th cells [11].
Figure 4. HLA class II antigen-processing and presentation pathway
5. Distribution of HLA
The distribution of HLA antigens in different ethnic groups has a unique pattern. Take HLA-B27 as an example, which is a gene associated with the prevalence of AS. The areas with higher B27 positive rates are mainly American Columbia, the Indian population along the coast of Canada, and Canadian Haida. The positive rate of B27 in other regions is as follows: It was 6%-8% in European and American Caucasians; less than 1% in Japan and Africa; in the Chinese population is 4% ~ 8%.
6. Polymorphism of HLA
The MHC gene is the most polymorphic gene in the human genome (total HLA allele 13023; HLA I allele 9749 and HLA II allele 3274) [12]. The specific information is shown in table 2 and table 3.
There are two main hypotheses about the polymorphism of HLA:
● The first view is that polymorphism is maintained by heterozygote dominance. Since HLA gene expression is codominant, HLA heterozygous expression of functional HLA protein types is absolutely more than homozygous expression.
● The second hypothesis is that HLA polymorphism is the result of frequency-dependent selection during evolution between pathogens and vertebrate immune systems.
The polymorphism of MHC, on the one hand, is an obstacle to finding a match, and on the other hand, it enables the immune system to recognize any invading pathogens.
Table 2 The number of alleles of HLA Class I
| HLA Class I |
| Gene |
A |
B |
C |
E |
F |
G |
| Alleles |
4,638 |
5,590 |
4,374 |
27 |
31 |
61 |
| Proteins |
3,172 |
3,923 |
2,920 |
8 |
6 |
19 |
| Nulls |
224 |
169 |
171 |
1 |
0 |
3 |
Table 3 The number of alleles of HLA Class II
| HLA Class II |
| Gene |
Alleles |
Proteins |
Nulls |
| DRA |
7 |
2 |
0 |
| DRB |
2,639 |
1,908 |
84 |
| DQA1 |
100 |
36 |
4 |
| DQB1 |
1,316 |
878 |
35 |
| DPA1 |
73 |
32 |
0 |
| DPA2 |
5 |
2 |
0 |
| DPB1 |
1,097 |
728 |
34 |
| DPB2 |
6 |
3 |
0 |
| DMA |
7 |
4 |
0 |
| DMB |
13 |
7 |
0 |
| DOA |
12 |
3 |
1 |
| DOB |
13 |
5 |
0 |
Data from IMGT - HLA database: https://www.ebi.ac.uk/ipd/imgt/hla/stats.html
7. Human Leukocyte Antigen (HLA) Test
HLA testing, also known as HLA typing or tissue typing, is a blood test that identifies antigens on the surface of cells and tissues. HLA genes play an important role in transplant rejection as well as infective and autoimmune diseases [13]. Accurate HLA typing is thus important both in clinical and research. It is required for the recipient to receive an HLA test before a donor stem cell or organ transplant to determine whether the donor's tissues match that of the recipient.
The more antigens that match, the higher the likelihood that a transplant will be successful. HLA compatibility lowers the risk of complications like graft-versus-host disease (GVHD) and facilitates the growth and development of engraftment. However, the ideal transplant option for some patients is a doctor who matches exactly half of their HLA, a phenomenon known as a haploidentical or half-matched transplant.
HLA typing has been done using a variety of techniques, such as serological, cellular, and molecular analysis [14]. With the birth of DNA sequencing and polymerase chain reaction (PCR), specific oligonucleotide probe hybridization, sequence-specific primer amplification, sequence typing (SBT), and other molecular typing techniques have been developed.
8. The Applications of HLA
The clinical application of HLA is mainly the matching of donor and recipient in organ transplantation [15]. HLA typing can be used to match patients and donors of bone marrow or cord blood transplantation. Besides, some HLA molecules can be used to diagnose certain diseases.
8.1 Transplant
In bone marrow transplantation, the donor and the recipient must have the same or matching HLA gene, so that the transplantation can be successful and the donor tissue cannot be attacked or rejected by the recipient's immune system.
In addition to the classical HLA-A, HLA-B, and HLA-DR antigens, the role of HLA-C and HLA-DQ antigens in graft survival or sensitization is now well documented [16][17].
In the hematopoietic stem cell transplantation, HLA molecular allelic typing is often performed to provide HLA class I and class II allele matching.
8.2 HLA and clinical blood transfusion
HLA is closely related to blood transfusion, mainly due to the immune response of HLA. Due to the high immunogenicity of HLA antigen, HLA antibodies can be produced by immunizing the body through pregnancy, blood transfusion, transplantation, and other ways. Heterologous HLA immunization causes various problems in transfusion therapy, such as platelet transfusion refractoriness (RTR), febrile non-hemolytic transfusion (FNHTR), transfusion-related acute lung injury (TRA-LI), Transfusion-associated graft-versus-host disease (TA-GVHD). In order to avoid these situations, HLA detection very necessary HLA detection is required for this situation.
HLA genotyping plays an important role in determining the immune compatibility between donor and recipient and is also part of the diagnosis of some autoimmune diseases.
8.3 Disease Diagnosis
HLA-B27 is a human leukocyte antigen and belongs to the MHC class I gene. In clinical work, B27 positive can help us diagnose ankylosing spondylitis (AS). However, the diagnosis of AS needs to be judged by combining clinical symptoms, physical examination, imaging examination, and laboratory examination. HLA-B27 alone cannot diagnose and exclude AS. Nevertheless, B27 still has an important auxiliary value for the diagnosis of AS.
Currently, there are four commonly used methods for B27 detection: flow cytometry, PCR-SSP, ELISA, and microcytotoxic assay. Flow cytometry is currently the most ideal method to detect B27, and it is also recommended internationally to detect B27.
9. HLA-Associated Diseases
Many HLA genes are linked to human diseases, including some autoimmune diseases and cancer. But some mechanism has not been fully explained.
9.1 HLA and Autoimmune Diseases
HLA-B alleles such as HLA-B* 2702 and HLA-B* 2705 are associated with ankylosing spondylitis (AS). HLA-DRB1 alleles including DRB1 * 04:01, DRB1 * 04:04 DRB1 *04:05, and DRB1 * 01:01) are related to seropositive rheumatoid arthritis (RA), HLA-DQB1*0602 is associated with narcolepsy [18]. Celiac disease is associated with HLA-DQB1*02. HLA-A1, B8, and DR17 haploids are often linked to autoimmune diseases. Type I diabetes is associated with HLA-DR3 [18]. Psoriasis is associated with HLA-C. Multiple sclerosis is associated with HLA-DR2. Rheumatoid arthritis is associated with HLA-DR4.
9.2 HLA and Infectious Diseases
Studies have confirmed the relationship between HLA molecules and resistance to falciparum malaria in West African populations [19]. Combined with statistical analysis, it is concluded that class I HLA-B*53 and class II DRB1*1302-DQB1*0501 haplotype in Gambia is closely related to susceptibility to falciparum malaria. Individuals with one of the two are less susceptible to falciparum malaria. In other words, these two molecules can reduce the incidence of severe infections. This discovery led to a major advance in the study of the relationship between HLA antigens and infectious disease immunity.
Additionally, many studies have shown that HLA alleles are associated with various aspects of AIDS. Existing evidence indicates that HLA is the most important site for human HIV differential control [20]. Kaslow et al. evaluated the role of HLA I alleles in HIV infection and found that HLA B27 and B57 were closely related to the slow progress of AIDS [21].
HLA homozygous individuals are more likely to become infected with HIV than HLA heterozygous individuals. This is because individuals heterozygous at HLA sites will be able to provide T cells with a wider pool of antigenic peptides than homozygous individuals, thus exerting greater pressure on pathogens.
9.3 HLA and Cancer
HLA expression is closely related to malignant tumors. The expression of classical HLA-I molecules is generally down-regulated in tumor cells, which is the selective escape mechanism of tumor cells to present tumor antigen peptides by HLA molecules. The lower the expression of the HLA-I molecule, the higher the malignancy of the tumor and the worse the prognosis of patients.
Zia et al. confirmed that the decreased expression of the HLA-I molecule was consistent with the malignant degree of breast cancer by flow cytometry, and proposed that the HLA-I molecule could be used as a reference index to reflect breast cancer metastasis [22]. Jamil's study showed that the expression of the heavy chain (HLA-A, HLA-B, HLA-C) and light chain antigen (β2-m) at various points of HLA-I molecules in colorectal cancer was significantly lower than that in normal mucosa [23].
The correlation between abnormal expression of HLA-II molecules and tumors is complex, mainly reflected in the expression of HLA-DR, but most of the situation is the same as that of class I molecules. HLA-II antigen can also be expressed on the surface of some tumor cells, which is closely related to the immunogenicity, metastasis, and prognosis of tumors.
Polymorphism of the HLA-I molecule is also associated with susceptibility to malignancy. HLA-I polymorphism causes significant differences differences in the conformation of antigen binding grooves and the efficiency of binding antigen-presenting peptides, resulting in different susceptibility of HLA genotypes to tumors. These susceptible tumors include ovarian cancer, cervical cancer, stomach cancer, and malignant melanoma.
9.4 HLA and Pregnancy
Problems that can occur in pregnant women: recurrent miscarriage, pre-eclampsia, or hemolytic disease in the newborn. These conditions are considered to be immune rejection.
Some mechanisms have been proposed to explain the immune privilege state of the diaphragm. Different assumptions can be grouped into five main points: the mechanical barrier effect of the trophoblast; inhibition of the maternal immune system during pregnancy; deletion of HLA class I molecules in trophoblasts; cytokine changes; local immunosuppression mediated by Fas/FasL system.
There is such an interesting phenomenon in fetal tissues: low HLA-C expression and lack of highly polymorphic HLA-A molecules.
10. HLA and Drug Sensitivity
Allergic drug reactions usually occur in low molecular weight drugs. Several explanations exist for the mechanism of a drug allergy associated with HLA.
Drugs and their metabolites are too small to produce their own immunogenicity, but they can be used as a hapten to modify certain autologous proteins in the host, leading to immune recognition of the hapten produced: self - peptide complex as a new antigen [24]. An example of drug haptens is penicillin, which has chemical activity and stable covalent binding with proteins or polypeptides to produce immunogenic self-proteins [25].
Drugs can bridge TCR and HLA molecules without directly binding to peptide antigens. Drug complexes can directly activate the immune response of T cells without the need for specific peptide ligands [26]. For example, sulfamethoxazole can bind non-covalently to antigen-presenting structures (such as TCR or HLA) and directly cause irritation of immune responses.
In conclusion, HLA is primarily important as an antigen-presenting molecule involved in adaptive immune responses and as a regulatory molecule participating in innate immune responses. The success or failure of organ transplantation is determined by the matching degree of HLA typing between donor and recipient. HLA typing is also associated with transfusion reactions caused by leukocyte antigens such as non-hemolytic transfusion reactions, mainly in patients receiving repeated transfusion therapy. Abnormal expression of HLA is responsible for a variety of diseases, including automatic immune diseases and cancer. HLA polymorphisms also reflect differences in susceptibility to disease.
As is widely known, CTLs play a significant role in destroying tumor cells and managing viral or bacterial infections, whereas CD4+ T cells are necessary for the priming and development of naive CD8+ T cells as well as the secondary expansion of CD8+ memory T cells. Therefore, it might be crucial to integrate HLA-I- and -II-restricted epitopes in peptide-based vaccines to get both CD4+ and CD8+ T cells for the induction of powerful and long-lasting immunity.
CUSABIO provides multiple HLA-associated products for HLA-related research, including recombinant proteins, antibodies, and ELISA kits. These products are used for scientific research only.
| HLA Antigens |
Product Name |
| HLA-A |
Recombinant Human HLA class I histocompatibility antigen, A alpha chain (HLA-A) |
| HLA-A Antibody |
| HLA-B |
Recombinant Human HLA class I histocompatibility antigen,B alpha chain (HLA-B) |
| HLA-B Antibody |
| HLA-B Antibody, HRP conjugated |
| HLA-B Antibody, FITC conjugated |
| HLA-B Antibody, Biotin conjugated |
| HLA-C |
Recombinant Human HLA class I histocompatibility antigen, Cw-7 alpha chain (HLA-C) |
| HLA-C Antibody |
| HLA-C Antibody, FITC conjugated |
| HLA-C Antibody, HRP conjugated |
| HLA-C Antibody, Biotin conjugated |
| HLA-D |
Recombinant Human HLA class II histocompatibility antigen, DQ alpha 1 chain (HLA-DQA1) |
| HLA-DQA1 Antibody |
| HLA-DQA1 Antibody, Biotin conjugated |
| HLA-DQA1 Antibody, FITC conjugated |
| HLA-E |
Recombinant Human HLA class I histocompatibility antigen, alpha chain E (HLA-E) |
| HLA-E Antibody |
| HLA-E Antibody, HRP conjugated |
| HLA-E Antibody, FITC conjugated |
| HLA-E Antibody, Biotin conjugated |
| Human major histocompatibility complex, class I, E (HLA-E) ELISA kit |
| HLA-F |
Recombinant Human HLA class I histocompatibility antigen, alpha chain F (HLA-F) |
| HLA-F Antibody |
| HLA-F Antibody, FITC conjugated |
| HLA-F Antibody, HRP conjugated |
| HLA-G |
Recombinant Human HLA class I histocompatibility antigen, alpha chain G (HLA-G) |
| HLA-G Antibody |
| HLA-G Antibody, Biotin conjugated |
| HLA-G Antibody, HRP conjugated |
| HLA-G Antibody, FITC conjugated |
| Human leukocyte antigen G, HLA-G ELISA Kit |
| HLA-DP |
Recombinant Human HLA class II histocompatibility antigen, DP alpha 1 chain (HLA-DPA1) |
| HLA-DPA1 Antibody |
| HLA-DPA1 Antibody, HRP conjugated |
| HLA-DQ |
Recombinant Human HLA class II histocompatibility antigen, DQ alpha 1 chain (HLA-DQA1) |
| Recombinant Human HLA class II histocompatibility antigen, DQ beta 2 chain (HLA-DQB2) |
| Recombinant Human HLA class II histocompatibility antigen, DQ alpha 2 chain (HLA-DQA2) |
| Recombinant Human HLA class II histocompatibility antigen,DQ beta 1 chain (HLA-DQB1) |
| HLA-DQA1 Antibody |
| HLA-DQA1 Antibody, Biotin conjugated |
| HLA-DQA1 Antibody, FITC conjugated |
| HLA-DQA2 Antibody |
| HLA-DQB1 Antibody |
| HLA-DQB1 Antibody, Biotin conjugated |
| HLA-DQB1 Antibody, FITC conjugated |
| HLA-DQB1 Antibody, HRP conjugated |
| HLA-DR |
Recombinant Human HLA class II histocompatibility antigen, DR alpha chain (HLA-DRA) |
| Recombinant Human HLA class II histocompatibility antigen, DRB1-15 beta chain (HLA-DRB1) |
| Recombinant Human HLA class II histocompatibility antigen, DR beta 5 chain (HLA-DRB5) |
| HLA-DRB5 Antibody |
| HLA-DRA Antibody |
| HLA-DRA Antibody, FITC conjugated |
| HLA-DRA Antibody, HRP conjugated |
| Human HLA class II histocompatibility antigen, DR alpha chain (HLA-DRA) ELISA kit |
| HLA-DM |
Recombinant Human HLA class II histocompatibility antigen, DM alpha chain (HLA-DMA) |
| Recombinant Human HLA class II histocompatibility antigen, DM beta chain (HLA-DMB) |
| HLA-DMA Antibody |
| HLA-DMA Antibody, Biotin conjugated |
| HLA-DMA Antibody, FITC conjugated |
| HLA-DMA Antibody, HRP conjugated |
| HLA-DMB Antibody |
| HLA-DO |
Recombinant Human HLA class II histocompatibility antigen, DO alpha chain (HLA-DOA) |
| Recombinant Human HLA class II histocompatibility antigen, DO beta chain (HLA-DOB) |
| HLA-DOA Antibody |
| HLA-DOB Antibody |
References
[1] Jr C A J, Travers P, Walport M, et al. The major histocompatibility complex and its functions - Immunobiology - NCBI Bookshelf [J]. Garland Science, 2001.
[2] Beck S, Trowsdale J. The human major histocompatibility complex: Lessons from the DNA Sequence [J]. Annu Rev Genomics Hum Genet, 2000, 1(1): 117-137.
[3] Salter RD, Benjamin RJ, Wesley PK, et al. A binding site for the T-cell co-receptor CD8 on the alpha 3 domain of HLA-A2 [J]. Nature. 1990;345:41–46.
[4] York IA, Rock KL. Antigen processing and presentation by the class I major histocompatibility complex [J]. Annu Rev Immunol. 1996;14:369–396.
[5] Vitiello A, Potter TA, Sherman LA. The role of beta 2-microglobulin in peptide binding by class I molecules [J]. Science. 1990;250:1423–1426.
[6] Perarnau B, Siegrist CA, Gillet A, et al. Beta 2-microglobulin restriction of antigen presentation [J]. Nature. 1990;346:751–754.
[7] Li XC, Raghavan M. Structure and function of major histocompatibility complex class I antigens [J]. Curr Opin Organ Transplant 2010;15:499–504.
[8] Cresswell P, Ackerman AL, Giodini A, et al. Mechanisms of MHC class I-restricted antigen processing and cross-presentation [J]. Immunol Rev. 2005;207:145–157.
[9] Kloetzel PM. Antigen processing by the proteasome [J]. Nat Rev Mol Cell Biol. 2001;2:179–187.
[10] Watts C. The exogenous pathway for antigen presentation on major histocompatibility complex class II and CD1 molecules [J]. Nat Immunol. 2004;5:685–692.
[11] Wagner C S, Grotzke J E, Cresswell P. Intracellular events regulating cross-presentation [J]. Frontiers in Immunology, 2012, 3: 138.
[12] Robinson J, Halliwell J A, Hayhurst J D, et al. The IPD and IMGT/HLA database: allele variant databases [J]. Nucleic Acids Research, 2015, 43(D1): D423-D431.
[13] Matzaraki V, Kumar V, Wijmenga C, et al. The MHC locus and genetic susceptibility to autoimmune and infectious diseases [J]. Genome Biology, 2017, 18(1): 76.
[14] Lind C, Ferriola D, Mackiewicz K, et al. Next-generation sequencing: the solution for high-resolution, unambiguous human leukocyte antigen typing [J]. Human Immunology, 2010, 71(10): 0-1042.
[15] Bhadran Bose D W J, Campbell S B. Transplantation Antigens and Histocompatibility Matching [J]. Intech, 2013.
[16] Tran T H , Döhler, Bernd, Heinold A, et al. Deleterious Impact of Mismatching for Human Leukocyte Antigen-C in Presensitized Recipients of Kidney Transplants [J]. Transplantation, 2011, 92(4): 419.
[17] Tambur A R, Leventhal J R, Friedewald J J, et al. The Complexity of Human Leukocyte Antigen (HLA)-DQ Antibodies and Its Effect on Virtual Crossmatching [J]. Transplantation, 2016, 90(10): 1117-1124.
[18] Holoshitz J. The quest for better understanding of HLA-disease association: scenes from a road less travelled by [J]. Discovery Medicine, 2013, 16(87): 93-101.
[19] Gilbert SC, Plebanski M, Gupta S, et al. Association of malaria parasite population structure, HLA, and immunological antagonism [J]. Science 1998;279:1173–7.
[20] Martin M P, Carrington M. Immunogenetics of HIV disease [J]. Immunological Reviews, 2013, 254(1): 245-264.
[21] Kaslow R A, Carrington M, Apple R, et al. Influence of combinations of human major histocompatibility complex genes on the course of HIV-1 infection [J]. Nature Medicine, 1996, 2(4): 405.
[22] Zia A, Schidberg FW, Funke I. MHC class I negative phenotype of disseminated tumor cells in bone narrow is associated with poor survival in ROMO breast cancer patients [J]. Int Cancer, 2001, 93(4):566.
[23] Jamil KA, Bharti O, Kanti DB. Immune responses in cancer [J]. J Pharmacol Therapeut, 2003,99:113-132.
[24] Yawalkar N, Pichler W J. Pathogenesis of Drug-Induced Exanthema [J]. International Archives of Allergy and Immunology, 2001, 124(1-3): 336-338.
[25] Warrington R. Drug allergy: causes and desensitization [J]. Human Vaccines & Immunotherapeutics, 2012, 8(10): 1513.
[26] Pichler W J, Beeler A, Keller M, et al. Pharmacological interaction of drugs with immune receptors: the p-i concept [J]. Allergology International, 2006, 55(1): 17-25.