[1] Wu, Cheng-Yu, et al. "CEACAM6 as a Novel Therapeutic Target to Boost HO-1-mediated Antioxidant Defense in COPD." American Journal of Respiratory and Critical Care Medicine 207.12 (2023): 1576-1590.
[2] Zang, Mingde, et al. "CEACAM6 promotes tumor angiogenesis and vasculogenic mimicry in gastric cancer via FAK signaling." Biochimica et Biophysica Acta (BBA)-Molecular Basis of Disease 1852.5 (2015): 1020-1028.
[3] Thomas, Jerin, et al. "CEACAMS 1, 5, and 6 in disease and cancer: interactions with pathogens." Genes & Cancer 14 (2023): 12.
[4] Blumenthal, Rosalyn D., et al. "Expression patterns of CEACAM5 and CEACAM6 in primary and metastatic cancers." BMC cancer 7 (2007): 1-15.
[5] Zang, Mingde, et al. "Dual role of carcinoembryonic antigen-related cell adhesion molecule 6 expression in predicting the overall survival of gastric cancer patients." Scientific Reports 7.1 (2017): 10773.
[6] Jantscheff, Peter, et al. "Expression of CEACAM6 in resectable colorectal cancer: a factor of independent prognostic significance." Journal of clinical oncology 21.19 (2003): 3638-3646.
[7] Blumenthal, Rosalyn D., Hans J. Hansen, and David M. Goldenberg. "Inhibition of adhesion, invasion, and metastasis by antibodies targeting CEACAM6 ( NCA-90) and CEACAM5 (Carcinoembryonic Antigen)." Cancer research 65.19 (2005): 8809-8817.
[8] Duxbury, Mark S., et al. "CEACAM6 is a novel biomarker in pancreatic adenocarcinoma and PanIN lesions." Annals of surgery 241.3 (2005): 491.
[9] Blumenthal R D, Leon E, Hansen H J, et al. Expression patterns of CEACAM5 and CEACAM6 in primary and metastatic cancers[J]. BMC cancer, 2007, 7: 1-15.
[10] Johnson, Benny, and Daruka Mahadevan. "Emerging role and targeting of carcinoembryonic antigen-related cell adhesion molecule 6 (CEACAM6) in human malignancies." Clinical cancer drugs 2.2 (2015): 100-111.
[11] Liu, Yingying, et al. "FOXP3 and CEACAM6 expression and T cell infiltration in the occurrence and development of colon cancer." Oncology Letters 11.6 ( 2016): 3693-3701.
[12] Pinkert, Jessica, et al. "T cell-mediated elimination of cancer cells by blocking CEACAM6-CEACAM1 interaction." Oncoimmunology 11.1 ( 2022): 2008110.
[13] Ieta, K., et al. "CEACAM6 gene expression in intrahepatic cholangiocarcinoma." British journal of cancer 95.4 (2006): 532-540.
[14] Blumenthal R D, Hansen H J, Goldenberg D M. Inhibition of adhesion, invasion, and metastasis by antibodies targeting CEACAM6 (NCA-90) and CEACAM5 (Carcinoembryonic Antigen)[J]. Cancer research, 2005, 65(19): 8809-8817.
[15] Chen, Jianmin, et al. "CEACAM6 induces epithelial-mesenchymal transition and mediates invasion and metastasis in pancreatic cancer." International journal of oncology 43.3 (2013): 877-885.
[16] Duxbury, Mark S., et al. "Overexpression of CEACAM6 promotes insulin-like growth factor I-induced pancreatic adenocarcinoma cellular invasiveness. " Oncogene 23.34 (2004): 5834-5842.
[17] Duxbury, Mark S., et al. "A novel role for carcinoembryonic antigen-related cell adhesion molecule 6 as a determinant of gemcitabine chemoresistance in pancreatic adenocarcinoma cells." Cancer research 64.11 (2004): 3987-3993.
[18] Cheng, Tsai-Mu, et al. "Single domain antibody against carcinoembryonic antigen-related cell adhesion molecule 6 (CEACAM6) inhibits proliferation, migration, invasion and angiogenesis of pancreatic cancer cells." European Journal of Cancer 50.4 (2014): 713-721.
[19] Balk-Møller, Emilie, et al. "A marker of endocrine receptor-positive cells, CEACAM6, is shared by two major classes of breast cancer: luminal and HER2- enriched." the american journal of pathology 184.4 (2014): 1198-1208.
[20] Rizeq, Balsam, Zain Zakaria, and Allal Ouhtit. "Towards understanding the mechanisms of actions of carcinoembryonic antigen-related cell adhesion molecule 6 in cancer progression." Cancer science 109.1 (2018): 33-42.
[21] Rodia, Maria Teresa, et al. "LGALS4, CEACAM6, TSPAN8, and COL1A2: Blood markers for colorectal cancer-validation in a cohort of subjects with positive fecal immunochemical test results." Clinical Colorectal Cancer 17.2 (2018): e217-e228.
[22] Jin, Chunhui, et al. "T cell immunity induced by a bivalent Salmonella-based CEACAM6 and 4-1BBL vaccines in a rat colorectal cancer model." Oncology letters 13.5 (2017): 3753-3759.
[23] Duxbury, Mark S., et al. "c-Src-dependent cross-talk between CEACAM6 and αvβ3 integrin enhances pancreatic adenocarcinoma cell adhesion to extracellular matrix components." Biochemical and biophysical research communications 317.1 (2004): 133-141.
[24] Zhang, Yunqiang, et al. "CEACAM6 promotes tumor migration, invasion, and metastasis in gastric cancer." Acta Biochim Biophys Sin 46.4 (2014): 283-290.
[25] Li, Yingmei, et al. "Comprehensive RNA analysis of CSF reveals a role for CEACAM6 in lung cancer leptomeningeal metastases." NPJ Precision Oncology 5.1 ( 2021): 90.
[26] Sheng-lian, L. A. I., M. A. O. Min, and H. U. A. N. G. Lin. "Expression and significance analysis of CEACAM6, miR-146b and S100A6 in thyroid cancer." Journal of Hebei Medical University 44.5 (2023): 531.
[27] Zhu, Rujian, et al. "Carcinoembryonic antigen related cell adhesion molecule 6 promotes the proliferation and migration of renal cancer cells through the ERK/AKT signaling pathway." Translational Andrology and Urology 8.5 (2019): 457.
[28] Kanderová, Veronika, Ondřej Hrušák, and Tomáš Kalina. "Aberrantly expressed CEACAM6 is involved in the signaling leading to apoptosis of acute lymphoblastic leukemia cells." Experimental hematology 38.8 (2010): 653-660.
[29] Lasa, Adriana, et al. "High expression of CEACAM6 and CEACAM8 mRNA in acute lymphoblastic leukemias." Annals of hematology 87 (2008): 205-211.
[30] Steiner, N., et al. "Levels of CEACAM6 in peripheral blood are elevated in patients with plasma cell disorders: a potential new diagnostic marker and a new therapeutic target?." Disease Markers 2019 (2019).
[31] Witzens-Harig, Mathias, et al. "Tumor cells in multiple myeloma patients inhibit myeloma-reactive T cells through carcinoembryonic antigen-related cell adhesion molecule-6." Blood, The Journal of the American Society of Hematology 121.22 (2013): 4493-4503.
[32] Zhang, Feng, et al. "ING5 inhibits cancer aggressiveness via preventing EMT and is a potential prognostic biomarker for lung cancer." Oncotarget 6.18 ( 2015): 16239.
[33] Strickland, Laura A., et al. "Preclinical evaluation of carcinoembryonic cell adhesion molecule (CEACAM) 6 as potential therapy target for pancreatic adenocarcinoma." The Journal of Pathology: A Journal of the Pathological Society of Great Britain and Ireland 218.3 (2009): 380-390.
[34] Riley, Christopher J., et al. "Design and activity of a murine and humanized anti-CEACAM6 single-chain variable fragment in the treatment of pancreatic cancer." Cancer research 69.5 (2009): 1933-1940.
[35] Pandey, Ritu, et al. "Carcinoembryonic antigen cell adhesion molecule 6 (CEACAM6) in Pancreatic Ductal Adenocarcinoma (PDA): an integrative analysis of a novel therapeutic target." Scientific reports 9.1 (2019): 18347.

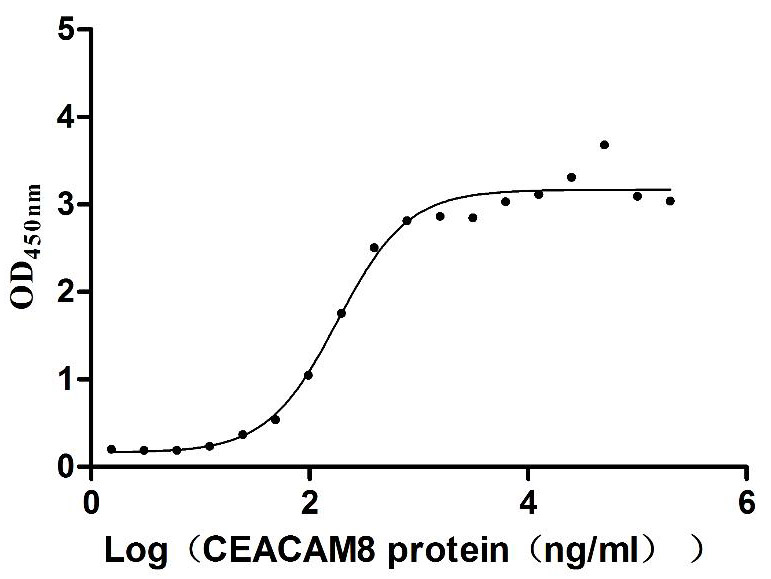
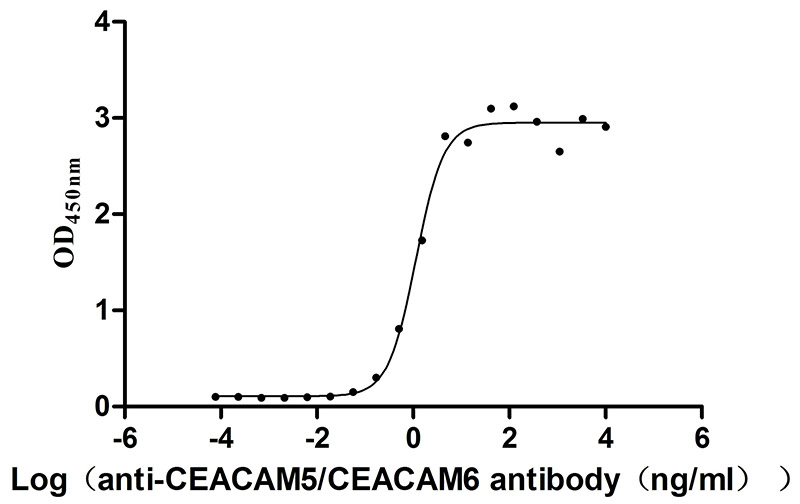
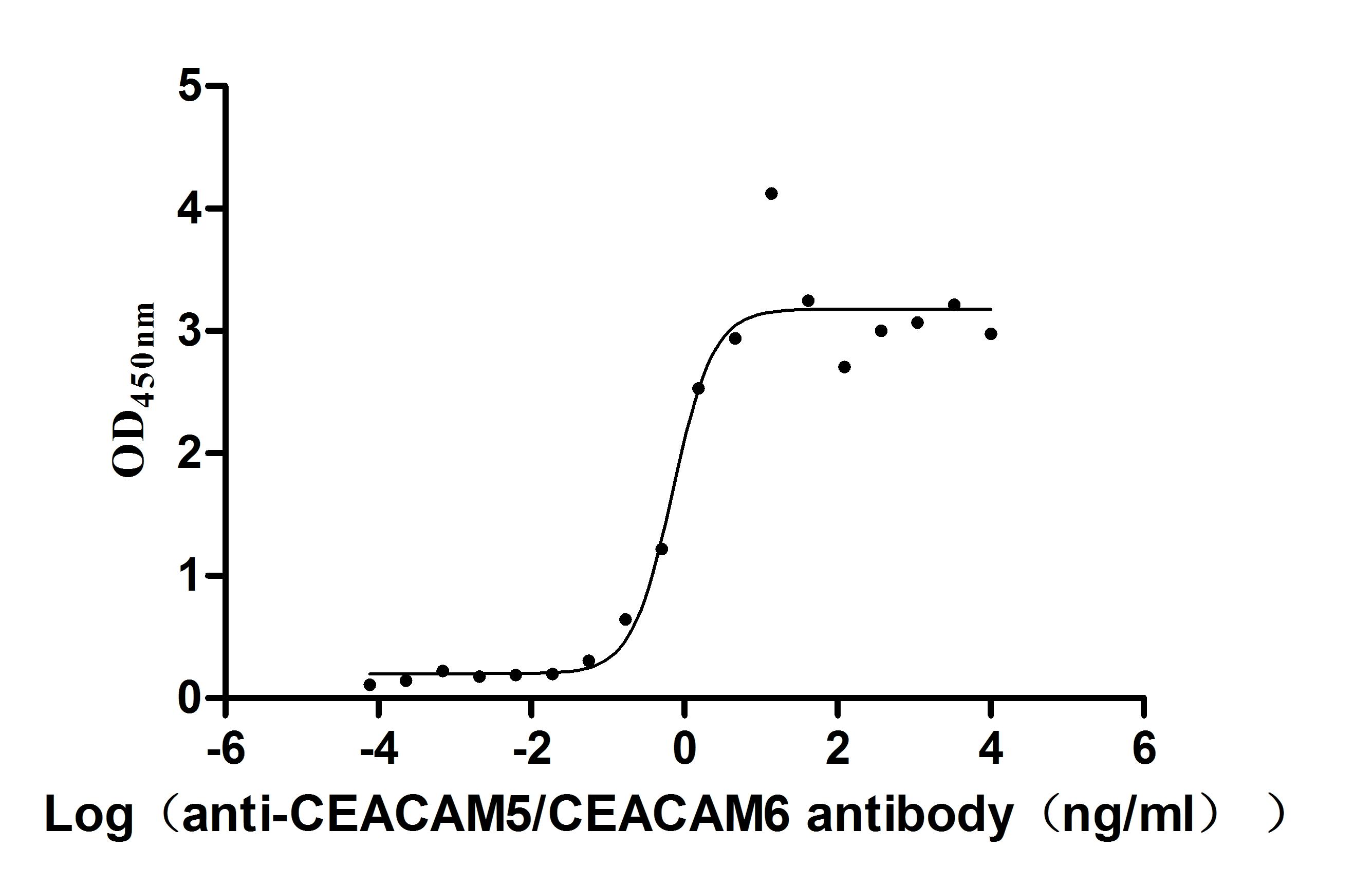
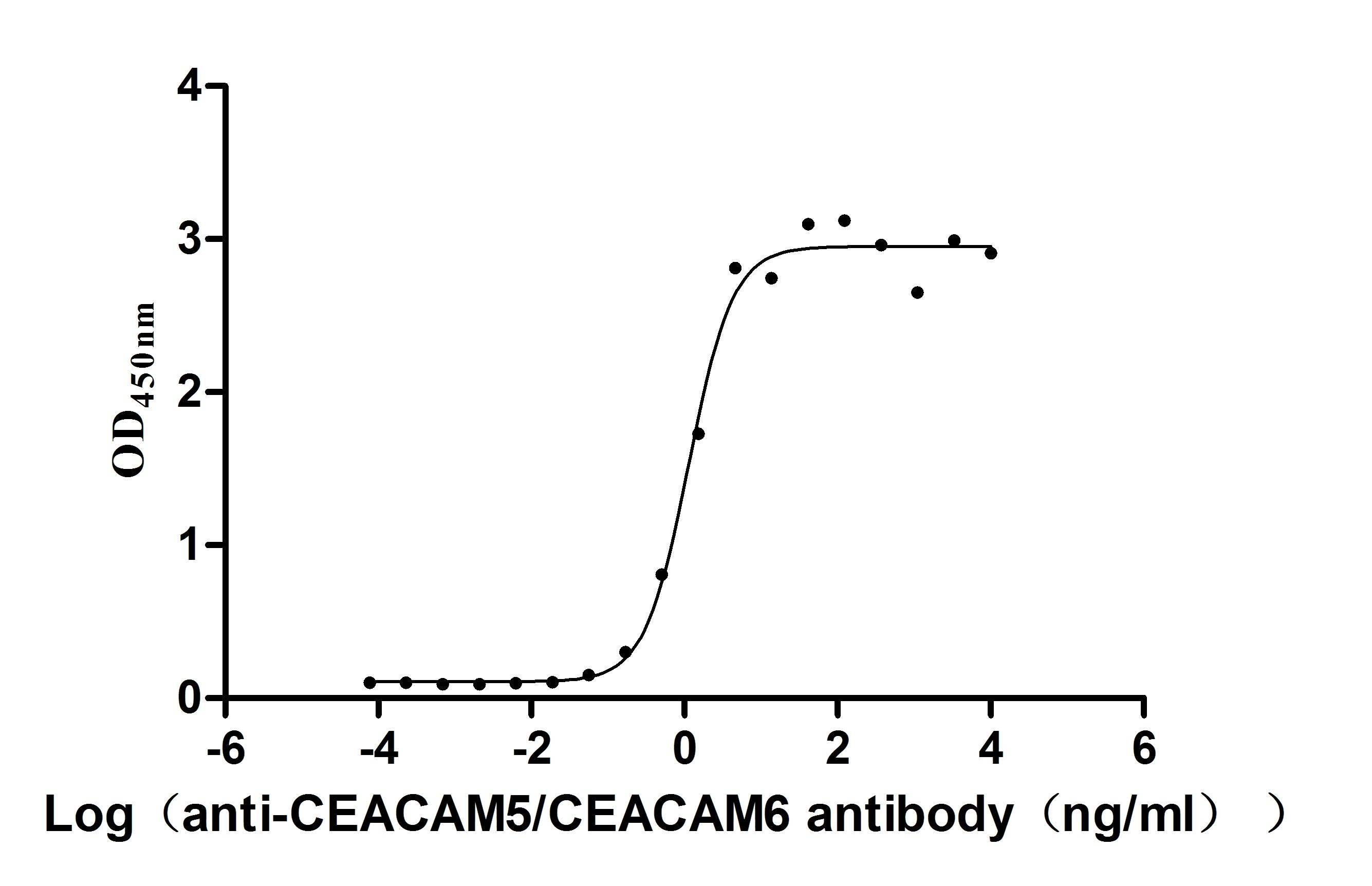


Comments
Leave a Comment