What Is Excitatory Synapse?
Synapses are the basic components of neural networks that allow information to be processed, encoded, and retrieved in the brain. They are composed of the presynaptic membrane, synaptic cleft, and postsynaptic membrane.
An excitatory synapse is a synapse that releases an excitatory neurotransmitter that brings the post-synaptic neuron membrane potential toward its threshold for firing an action potential. At the excitatory synapse, the neurotransmitters released into the synaptic cleft cause an action potential in the postsynaptic neuron.
Mechanisms of Excitatory Synapse Maturation
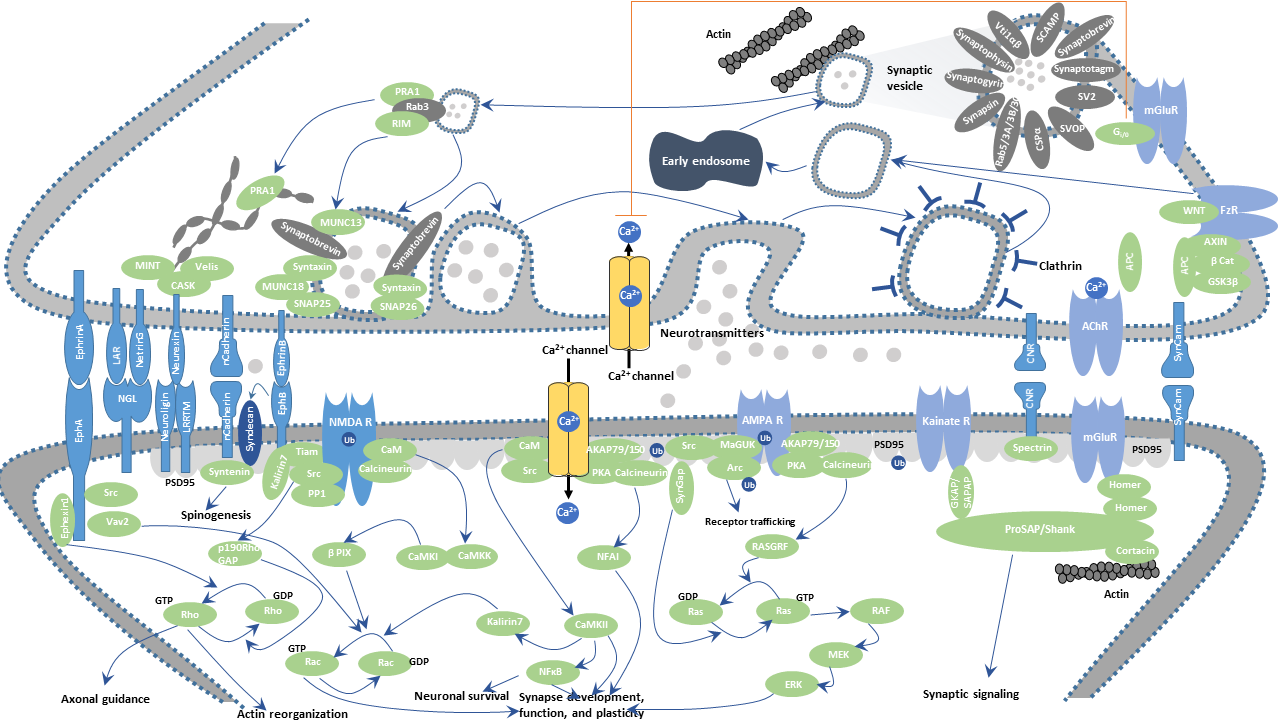
During development, the excitatory synapse is formed by signaling between presynaptic and postsynaptic neurons, where several classes of scaffolding proteins and adhesion molecules serve as inductive signals that trigger the establishment of presynaptic and postsynaptic specializations. This process includes contact formation between glutamate-releasing presynaptic terminals and postsynaptic membranes containing glutamate receptors and glutamate receptor clustering.
Excitatory synaptic transmission in the brain is predominantly mediated by the neurotransmitter glutamate. Glutamate is the primary excitatory neurotransmitter in the central nervous system (CNS) and is recycled from glutamine, produced by glial cells in the glutamate-glutamine cycle. An action potential induces the discharge of glutamate from the presynaptic terminals into the synaptic cleft. Specifically speaking, glutamate is packed into synaptic vesicles by a Ca2+/ATP-dependent transport process. Synaptic vesicles are docked at the active zone and released into the synaptic cleft from presynaptic sites by the exocytosis by the PRA1-Rab3-RIM complex. Upon binding to glutamate receptors including AMPARs, NMDARs, kainate receptors, and mGluRs, on the postsynaptic membranes, glutamate excites postsynaptic neurons by bringing in sodium ions and releasing potassium ions. The sodium ions inflow into the cell and the potassium ions exit the cell, leading to a reversal of the charges inside and outside the cell, making the postsynaptic neuronal cells depolarized, thus eliciting an action potential, known as excitatory postsynaptic potential (EPSP). Excess glutamate in the synaptic cleft is taken up by glial cells through EAAT1 and EAAT2 transporters and then is reconverted to glutamine, which is subsequently catalyzed into glutamate, resulting in a glutamine-glutamate cycle.
Presynaptic neurexin interacts with the Cbln1-GluD2 complex to trigger synapse formation. Neurexin interacts with the postsynaptic Neuroligin-1 (NLG1), recruiting NMDA receptors at a nascent synapse. Subsequent interaction of Neurexin with LRRTM2 results in the recruitment of AMPA receptors. Both cascades take place at the sites containing PSD-95. PSD-95 is a scaffolding molecule enriched at glutamatergic synapses and regulates the clustering of several neurotransmitter receptors, adhesion molecules, ion channels, cytoskeletal elements, and signaling molecules at postsynaptic sites.
The ephrins and Eph receptors are axon guidance and signaling molecules that are increasingly appreciated for their roles in synapse formation and maturation. For example, postsynaptic EphB2 influences AMPA receptor localization through PDZ binding domain interactions. Ephrin B2 interacts with GRIP to recruit and maintain AMPA receptors at synapses, while ephrin B3 is capable of strengthening excitatory synaptic transmission. EphA5-ephrin A5 interaction induces expression of PSD-95-NMDA receptor complexes and morphological spine maturation.
Leukocyte common antigen-related (LAR) appears to be co-opted at the synapse for specialization and development. Presynaptic LAR family protein tyrosine phosphatase receptors (PTRPs) signal through postsynaptic netrin-G ligand NGL-3 to induce excitatory synapse development.
The Function of the Excitatory Synapse Pathway
Synaptic excitation mediates a broad spectrum of structural changes in neural circuits across the brain. Excitatory synapses have a fundamental role in information processing within the brain and throughout the peripheral nervous system. At the excitatory synapses, glutamate interaction with glutamate receptors plays an important role in neuronal development and survival, synaptic plasticity, excitotoxicity, pain perception, and learning and memory.
Excitatory Synapses and Diseases
Excitotoxicity occurs when neurons are exposed to high quantities of glutamate that cause an exacerbated or prolonged activation of the NMDA and AMPA receptors and voltage-gated calcium channels leading to a lethal influx of extracellular calcium. Excitotoxicity starts a cascade of neurotoxicity that ultimately leads to the loss of neuronal function and cell death. Glutamate receptors have been associated with various neurological disorders. Many neurodegenerative diseases have been linked to glutamate receptor gene mutations or receptor autoantigen/antibody activity.



