What Are Focal Adhesions?
Focal adhesions (FAs) are adhesive sites where cells connect to the extracellular matrix (ECM). They contain clusters of transmembrane integrin receptors tethered at one end to the extracellular matrix (ECM) and the other to actin stress fibers, which are responsible for cell traction and ECM reorganization.
Focal Adhesion and Hemidesmosome
Focal adhesion mediates the adhesion between cells and the extracellular matrix. Hemidesmosomes anchor the epidermal keratin filament cytoskeleton to the extracellular matrix. They are crucial for the mechanical integrity of the skin.
The Function of Focal Adhesions
Focal adhesion is the physical action of the tight connection between the cells and the extracellular matrix, which enables cells to communicate with the external environment, causing cell adhesion, migration, diffusion, differentiation, and apoptosis.
The Mechanism of Focal Adhesions
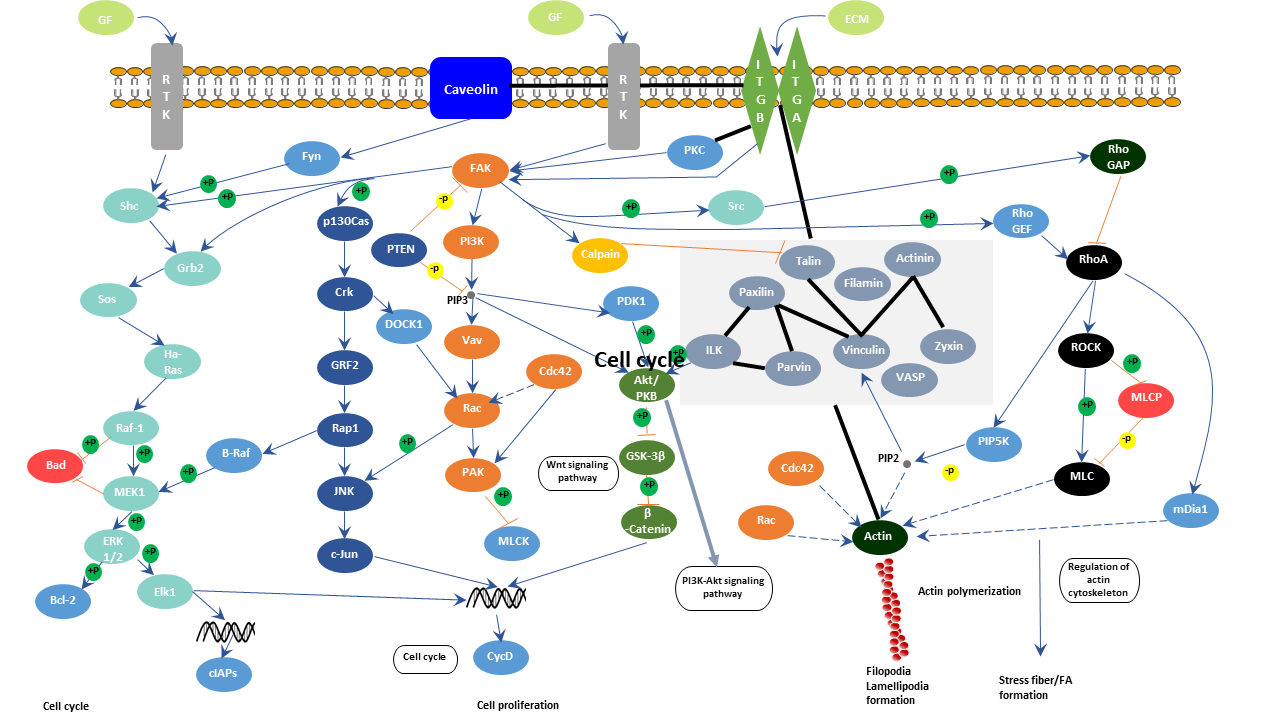
Focal adhesion is also called cell-matrix adhesion. Integrin receptors are the core of focal adhesions. Integrins span the plasma membrane and connect different extracellular matrix components. Integrins form heterodimers containing an alpha and beta subunit. The heterodimers bind to the extracellular matrix at its extracellular ligand-binding domain and anchors to the actin cytoskeleton at its cytosolic domain. Talin and alpha-actinin bind to the integrin's cytosolic domain. And the binding of talin with integrin helps recruit vinculin, paxillin, and focal adhesion kinase to cell-matrix adhesions. Vinculin usually cannot bind to other components because its binding site is automatically inhibited by the interaction between the head and tail domain. However, talin/alpha-actinin alone binding or in synergy with PIP2 disrupts the interaction and recovers vinculin's activity. Active vinculin subsequently links to cell-matrix adhesions through talin. Besides, vinculin also promotes the crosslink between the actin cytoskeleton and the integrin receptors by combining with actin filaments. This allows signaling transduction and communication between the extracellular matrix and cells. Therefore, this bidirectional force conduction from the inside to the outside of the cell and from the outside to the inside of the cell can control signal transmission in the intracellular or extracellular matrix through integrin conduction.
Diseases and Focal Adhesions
Cells continuously receive mechanical signals from the extracellular environment through adhesive spots. And cells respond to these signals through integrin-related signaling pathways to maintain cell morphology, migration, and survival. Thus, changes in the extracellular microenvironment that lack tone-regulation can lead to cancer progression and metastasis.
Nowadays, tumor metastasis and tumor therapeutic resistance are urgent problems in tumor research. Cancer treatment in the process of cell adhesion mediated radiation resistance (CAMRR) and cell adhesion mediated drug resistance (CAMDR) molecular mechanism research has certain progress. Several studies have shown that integrin as a cell receptor is important for the interaction between cells and the extracellular matrix environment in radiotherapy-acquired resistance phenotypes.



