What is EPHA3?
EphA3 (formerly known as HEK) is a type I transmembrane glycoprotein member of the Eph receptor family. Eph receptor family (erythropoietin- producing human hepatocellular carcinoma) is by far the largest RTKs family, has the function of regulating cell growth, differentiation and migration. Fourteen members of the Eph receptor family have been identified. According to their homology, expression, distribution and binding characteristics with ligands, they can be divided into two subgroups: EphA (EphA1, EphA2, EphA3, EphA4, EphA5, EphA6, EphA7, EphA8, EphA10, and EphB (EphB1, EphB2, EphB3, EphB4, EphB6).
EphA3 receptor was originally a surface antigen isolated from the surface cell line (LK63) of pre-B lymphoid leukemia cells. Its abnormal changes may lead to changes in cell morphological and biological characteristics, such as cell growth and survival rate, cell adhesion, cell migration and anti-apoptosis ability. The mutation of EphA3 is closely related to the occurrence and development of many kinds of tumors.
The Structure of EPHA3
EphA3 gene is located in 3p11.2. The protein it encodes includes three domains: extracellular ligand binding domain, hydrophobic transmembrane domain and intracellular tyrosine kinase active domain.
The extracellular ligand binding domain consists of a globular domain (Glb), a Sushi domain, an EGF-like domain and two fibronectin type Ⅲ repeats. The globular domain is a key domain that binds to ligands.
The intracellular domain includes tyrosine kinase active domain, SAM domain (Sterile α-motif domain) and PDZ domain. The SAM domain is highly conserved in the Eph protein family, and the tyrosine residue in the SAM structure is essential for the aggregation of receptor signal molecules.
EPHA3 Ligand
Ligands of EphA3: Ephrin-B2 and Ephrin-A5. Ephrin-B2 belongs to Ephrin-B subtype and belongs to single transmembrane protein, which is composed of intracellular domain, transmembrane domain and extracellular domain. Ephrin-A5 belongs to Ephrin-A subtype and is anchored to the cell membrane by glycosylphosphatidylinositol chain.
The Expression of EPHA3
EphA3 is highly expressed at all stages of embryonic development in brain, spinal cord, lung, kidney, heart and muscle tissue. EphA3 is almost nonexistent in normal adult tissues.
EphA3 is abnormally expressed in solid tumors such as gastric cancer, lung cancer, renal cell carcinoma, colon cancer, melanoma, sarcoma, bile duct cancer and prostate cancer, and is related to tumor invasion and metastasis. In addition, EphA3 is overexpressed in some hematopoietic tumors and lymphocytic tumors.
The Function of EPHA3
EphA3 receptors and ligands play an important role in normal cell adhesion, migration and angiogenesis. Eph-ephrin can also affect cell development by regulating cell-cell adhesion and de-adhesion to control cell localization during normal and carcinogenic development. Ephs is essential for the embryological process, especially for the development of the nervous system.
In addition, EphA3 receptors are closely related to the angiogenesis of many tumors and are thought to be involved in tumor invasion and metastasis. EphA3 is expressed in a variety of tumors and is related to tumor stem cells and angiogenesis. EphA3 has a wide range of mutations in cancer, and up to 40 mutations have been found in solid tumors.
The abnormal expression of EphA3 was also found in hematological malignancies. Eph RTKs is involved in the changes of tumor microenvironment, which may promote the occurrence of malignant tumors and angiogenesis.
More Interesting read related EPHA3>>
Popular Product
Recombinant Human Ephrin type-A receptor 3(EPHA3),partial (Active) (CSB-MP007723HU)
Validated Data
Purity determined
by
SDS-PAGE

Greater than 95% as determined by SDS-PAGE.
The binding activity of EFNA5-EPHA3 validated by functional ELISA
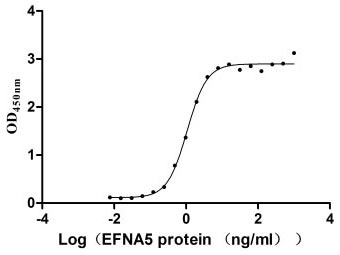
Immobilized EPHA3 at 2 μg/ml can bind human EFNA5 (CSB-MP007464HU), the EC50 of the protein is 0.9734-1.179 ng/ml.
The binding activity of EFNA5-EPHA3 validated
by LSPR
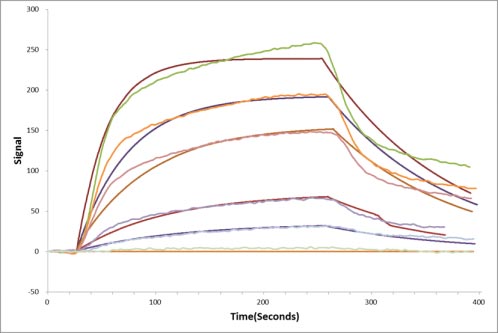
Human EPHA3 protein captured on COOH chip can bind Human EFNA5 protein with an affinity constant of 13.8 nM as detected by LSPR Assay.





