[1] Saxena, Richa, et al. "Genetic variation in GIPR influences the glucose and insulin responses to an oral glucose challenge." Nature genetics 42.2 (2010): 142-148.
[2] Canivell Fusté, Silvia. "The association of DNA methylation patterns in TCF7L2 and GIPR genes with Type 2 Diabetes." (2014).
[3] Sauber, Jeannine, et al. "Association of variants in gastric inhibitory polypeptide receptor gene with impaired glucose homeostasis in obese children and adolescents from Berlin." European journal of endocrinology 163.2 (2010): 259-264.
[4] Sauber, Jeannine, et al. "Association of variants in gastric inhibitory polypeptide receptor gene with impaired glucose homeostasis in obese children and adolescents from Berlin." European journal of endocrinology 163.2 (2010): 259-264.
[5] Erfanian, Saiedeh, et al. "Association of gastric inhibitory polypeptide receptor (GIPR) gene polymorphism with type 2 diabetes mellitus in iranian patients." BMC Medical Genomics 16.1 (2023): 44.
[6] El, K., et al. "GIP mediates the incretin effect and glucose tolerance by dual actions on α cells and β cells." Science advances 7.11 (2021): eabf1948.
[7] Suzuki, Kazuyo, et al. "Transcriptional regulatory factor X6 (Rfx6) increases gastric inhibitory polypeptide (GIP) expression in enteroendocrine K-cells and is involved in GIP hypersecretion in high fat diet-induced obesity." Journal of Biological Chemistry 288.3 (2013): 1929-1938.
[8] Ikeguchi, Eri, et al. "Transcriptional factor Pdx1 is involved in age-related GIP hypersecretion in mice." American Journal of Physiology-Gastrointestinal and Liver Physiology 315.2 (2018): G272-G282.
[9] Jepeal, Lisa I., Michael O. Boylan, and M. Michael Wolfe. "Cell-specific expression of the glucose-dependent insulinotropic polypeptide gene functions through a GATA and an ISL-1 motif in a mouse neuroendocrine tumor cell line." Regulatory peptides 113.1-3 (2003): 139-147.
[10] Kim, Su-Jin, Cuilan Nian, and Christopher HS McIntosh. "GIP increases human adipocyte LPL expression through CREB and TORC2-mediated trans-activation of the LPL gene." Journal of lipid research 51.11 (2010): 3145-3157.
[11] Ast, Julia, Johannes Broichhagen, and David J. Hodson. "Reagents and models for detecting endogenous GLP1R and GIPR." EBioMedicine 74 (2021): 103739.
[12] Mayendraraj, Ashok, Mette M. Rosenkilde, and Laerke S. Gasbjerg. "GLP-1 and GIP receptor signaling in beta cells–A review of receptor interactions and co-stimulation." Peptides (2022): 170749.
[13] Lavine, Jeremy A., and Alan D. Attie. "Gastrointestinal hormones and the regulation of β‐cell mass." Annals of the New York Academy of Sciences 1212.1 (2010): 41-58.
[14] Lyu, Xiaorui, et al. "Safflower yellow and its main component hydroxysafflor yellow A alleviate hyperleptinemia in diet‐induced obesity mice through a dual inhibition of the GIP‐GIPR signaling axis." Phytotherapy Research (2023).
[15] Dowsett, Georgina KC, et al. "A survey of the mouse hindbrain in the fed and fasted states using single-nucleus RNA sequencing." Molecular metabolism 53 (2021): 101240.
[16] Campbell, Jonathan E., et al. "TCF1 links GIPR signaling to the control of beta cell function and survival." Nature medicine 22.1 (2016): 84-90.
[17] Erfanian, Saiedeh, et al. "Association of gastric inhibitory polypeptide receptor (GIPR) gene polymorphism with type 2 diabetes mellitus in iranian patients." BMC Medical Genomics 16.1 (2023): 44.
[18] Zhang, Qian, et al. "The glucose-dependent insulinotropic polypeptide (GIP) regulates body weight and food intake via CNS-GIPR signaling." Cell metabolism 33.4 (2021): 833-844.
[19] Kee, Vanessa Ting Zhen. Dual incretin agonism in the treatment of Type 2 Diabetes and Obesity. Diss. Monash University, 2022.
[20] Bastin, Marie, and Fabrizio Andreelli. "Dual GIP–GLP1-receptor agonists in the treatment of type 2 diabetes: a short review on emerging data and therapeutic potential." Diabetes, Metabolic Syndrome and Obesity: Targets and Therapy (2019): 1973-1985.
[21] Davies, Iona, and Tricia MM Tan. "Design of novel therapeutics targeting the glucose-dependent insulinotropic polypeptide receptor (GIPR) to aid weight loss." Expert Opinion on Drug Discovery (2023): 1-11.
[22] Dekker, Mark J., et al. "Exercise prior to fat ingestion lowers fasting and postprandial VLDL and decreases adipose tissue IL-6 and GIP receptor mRNA in hypertriacylglycerolemic men." The Journal of nutritional biochemistry 21.10 (2010): 983-990.
[23] Campbell, Jonathan E. "Targeting the GIPR for obesity: to agonize or antagonize? Potential mechanisms." Molecular metabolism 46 (2021): 101139.
[24] Dalle Nogare, Mattia, et al. "The Methylation Analysis of the Glucose-Dependent Insulinotropic Polypeptide Receptor (GIPR) Locus in GH-Secreting Pituitary Adenomas." International Journal of Molecular Sciences 24.11 (2023): 9264.
[25] Sherman, Scott K., et al. "Gastric inhibitory polypeptide receptor (GIPR) is a promising target for imaging and therapy in neuroendocrine tumors." Surgery 154.6 (2013): 1206-1214.
[26] Sherman, S. K., et al. "High Expression of Somatostatin Type 2 Receptor (SSTR2) and Gastric Inhibitory Polypeptide Receptor (GIPR) in Stomach and Duodenal Neuroendocrine Tumors (NETs)." The Journal of surgical research 186.2 (2014): 506-506.
[27] Wang, Donghong, et al. "GIPR Expression in Gastric and Duodenal Neuroendocrine Tumors." (2014).

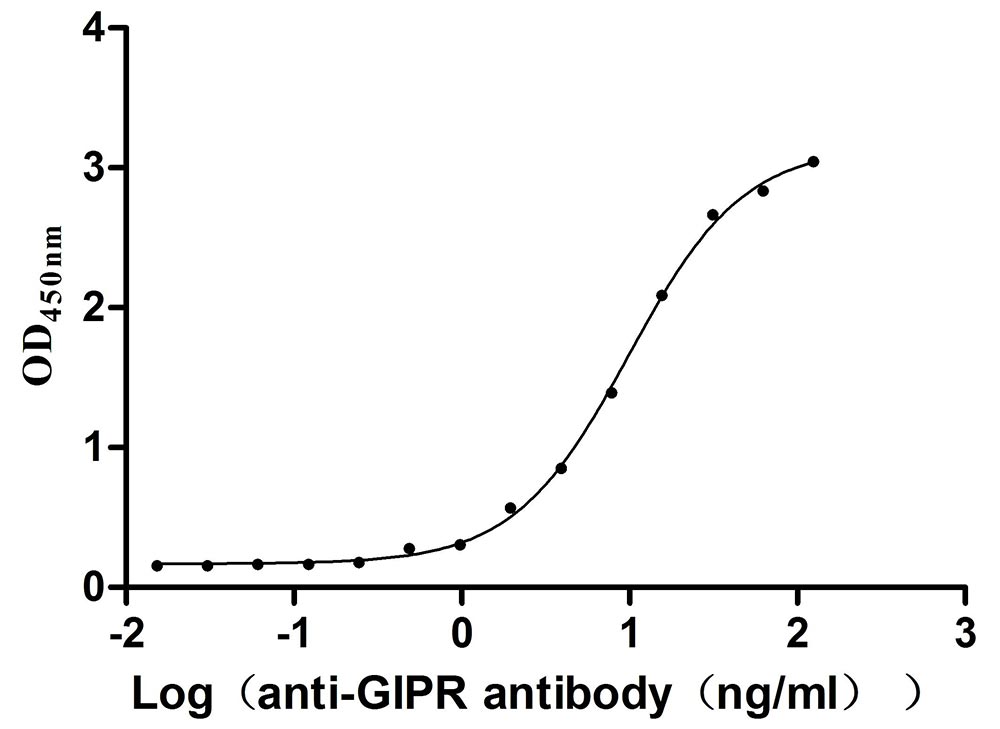
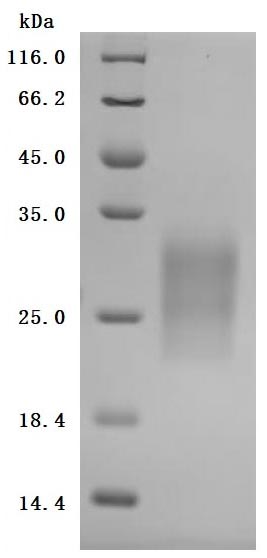
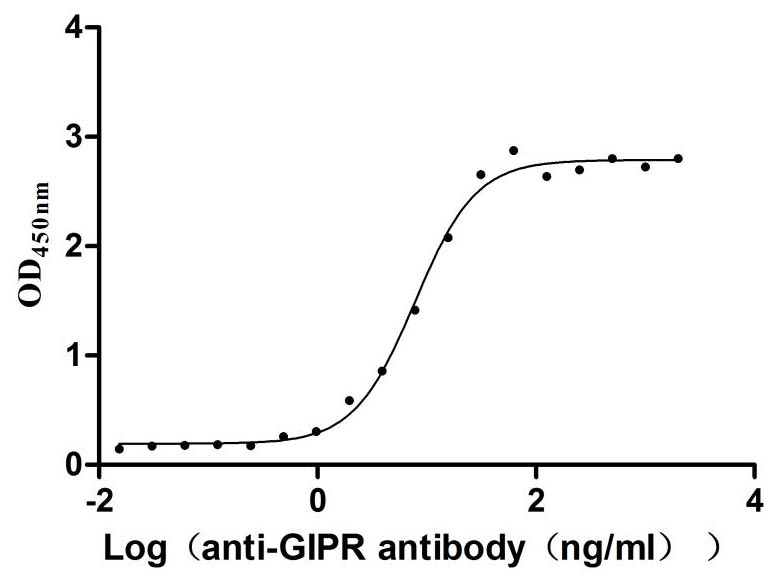
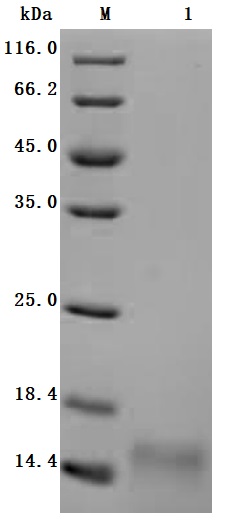


Comments
Leave a Comment