What is the Biological Role of Calcium Ion?
Calcium ion (Ca2+) serves as a universal second messenger in all eukaryotes. Ca2+ exerts a broad and wide range of biological roles inside and outside cells, such as muscle contraction, cell movement (the movement of flagella and cilia), neuronal transmission in the excitatory synapse, fertilization, neurogenesis, and cell growth or proliferation. And Ca2+ can also regulate the activity of enzymes (as a cofactor) & ion pumps and the permeability of ion channels. Moreover, it participates in the buildup of the cytoskeleton.
What is Calcium Signaling Pathway?
When neurotransmitters, hormones, growth factors, or initial chemical stimulation of excitatory cells stimulate cell membrane excitation, intracellular Ca2+ increases and then activates the Ca2+ signaling pathway. The Ca2+ signaling pathway consists of a series of molecular biological events that link external stimuli to appropriate responses within cells.
The Process of Calcium Signaling Pathway
The importance of Ca2+ signaling pathway for the implementation of the information provided by Ca2+has been increasingly appreciated and several distinct families of Ca2+ sensing proteins have been identified and characterized.
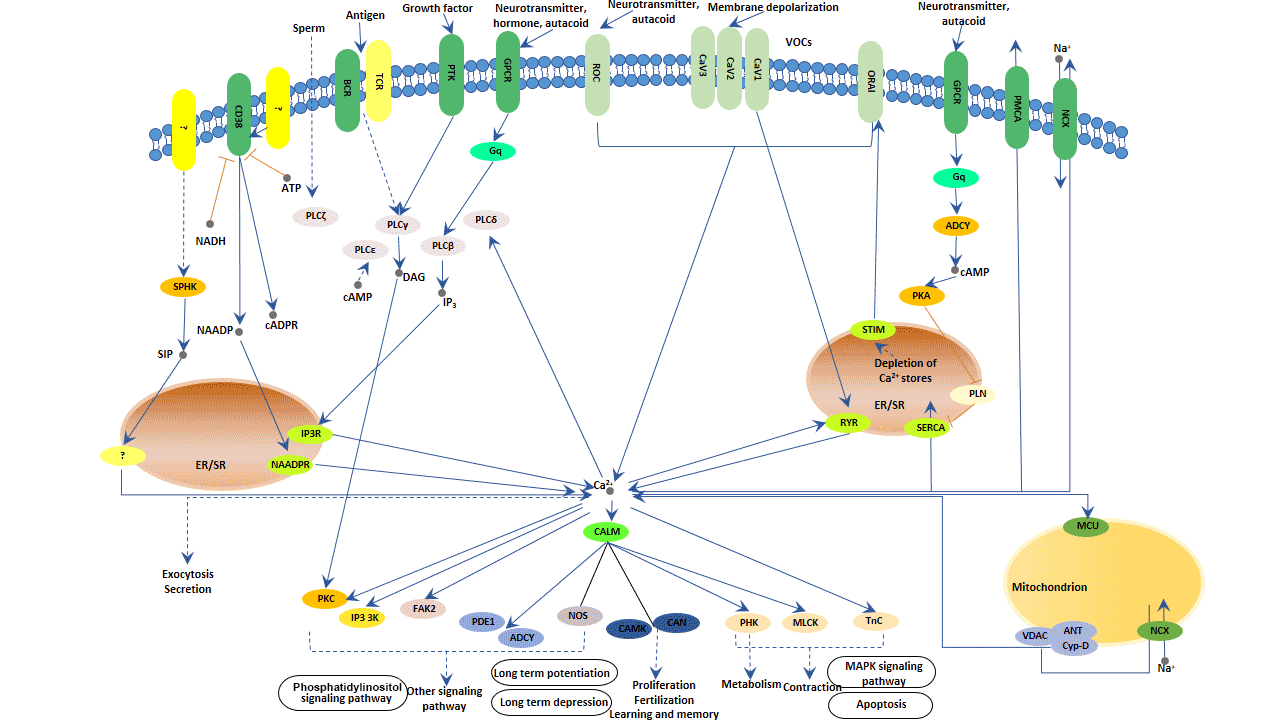
As it shows in the following picture, there are two principal sources of Ca2+ for signaling. One principal source of signal Ca2+ is the Ca2+ that enters the cell from the outside. The emergence of a large electrochemical gradient across the plasma membrane drives the entry of Ca2+. Cells use this external source of signal Ca2+ by activating various entry channels with widely different properties. The voltage-operated channels (VOCs) are found in excitable cells and generate the rapid Ca2+ fluxes that control fast cellular processes. Besides, there are also many other Ca2+-entry channels, including the receptor-operated channels (ROCs), second-messenger-operated channels (SMOCs) and store-operated channels (SOCs).
Another principal source of Ca2+ signaling comes from internal Ca2+ store that is primarily located in the endoplasmic/sarcoplasmic reticulum (ER/SR). And inositol-1,4,5-trisphosphate receptors (IP3Rs) or ryanodine receptors (RYRs) regulate the release of Ca2+ in ER/SR. The primary activator of these channels is Ca2+ itself. The process induced by Ca2+ will release Ca2+, which is critical to the mechanism of Ca2+ signaling. Various second messengers or modulators also control the release of Ca2+. In the phosphatidylinositol signaling pathway, phospholipase C (PLC-β) binds to hormones and the G protein-coupled receptors (GPCR) and then is activated. Activated PLC-β hydrolyzes 4, 5-bisphosphate phosphatidylinositol (PIP2) to form IP3. And IP3 regulates the IP3Rs. Cyclic ADP-ribose (cADPR) releases Ca2+ via RYRs. Nicotinic acid adenine dinucleotide phosphate (NAADP) may activate a distinct mechanism of Ca2+ release on separate acidic Ca2+ store. Ca2+ released from the NAADP-sensitive mechanism may also give feedback to either RYRs or IP3Rs.
The Function of Calcium Signaling Pathway
Only free calcium can play a role in the life activities within cells. Ca2+ plays a major coordinating role in almost all cellular processes. Ca2+ is a very important second messenger in cells. Many hormones and neurotransmitters achieve downstream signal transduction through the activation of calcium signaling by intracellular Ca2+ elevation, further causing gene expression, proliferation, substance secretion, motility, muscle contraction, the release of neurotransmitters, fertilization and apoptosis.
The Applications of Calcium Signaling Pathway
Calcium signaling has been widely demonstrated to be involved in plant responses to stresses. At present, it has been found that many kinds of stresses, such as low temperature, high temperature, oxygen stress, pathogenic factors, hypoxia, osmotic stress, and heavy metal ion, can cause the increase of cytoplasmic Ca2+ concentration. And many downstream stress reactions, like the changes in the expression of some genes, have also been proved to be related to the generated calcium signaling.
Ca2+ is not only an inducer for long-term enhancement (LTP) but also contributes to the maintenance of LTP. Ca2+ signals to activate downstream protein kinases and immediate early genes (IEGs) that promotes gene transcription and protein synthesis, thus leading to the phosphorylation of ERK. The phosphorylation of ERK is a key step in maintaining LTP.
Movement of Ca2+ from the extracellular compartment to the intracellular compartment alters membrane potential. This occurs in the heart during the plateau phase of ventricular contraction. In this example, Ca2+ acts to maintain the depolarization of the heart. Calcium signaling through ion channels is also important in neuronal synaptic transmission.
Besides, studies have proved that the Ca2+ signaling pathway plays an important role in invasion and metastasis of tumors cells.



