Necrosis
Necrosis, a type of cell death, refers to Irreversible cell injury and eventual cell death caused by various injurious stimulus, involving hypoxia, physical agents (trauma, extremes of temperature, radiation exposure, etc.), chemical agents (poisons, occupational exposure, drug toxicities, etc.), biological agents(bacteria, viruses, etc.), immunologic reactions. Cell injury can range from external injury to internal abnormalities [1] [2].
1. What are the Features and Types of Necrosis?
Comparing to apoptosis, necrosis is characterized by these features:
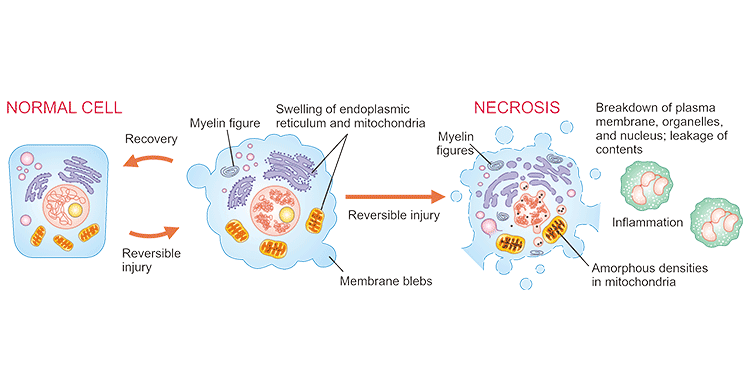
Necrosis is almost uncontrolled and release lots of chemicals from the dying cell to damage surrounding cells. Inflammation is often caused by necrosis. It occurs less frequently, involves many cells, may not be localized. It is a type of caspase independent cell death caused by external and internal injuries. Morphologically, necrotic cells characteristically demonstrate by swelling of organelles, such as the mitochondria and endoplasmic reticulum, the rupture of the plasma membrane, and the lysis of the cell [3].
Figure 1. Diagram of necrosis of a cell
In fact, There are many types of morphological patterns that necrosis can present itself, including coagulative, liquefactive, caseous, gangrenous, fat and fibrinoid necrosis.
In coagulative necrosis, the cell architecture remains preserved. Under the microscope, the cells appear anucleate, eosinophilic, with preserved structure. Liquefactive necrosis occurs in the central nervous system. In liquefactive necrosis, the dying cells are digested by hydrolytic enzymes and hence lose their structural integrity and turn into a viscous mass. Caseous necrosis is most commonly observed in tuberculous infection and refers to the whitish appearance of the necrotic area. Gangrenous necrosis is a clinical term for ischemic necrosis of the limbs, which is divided into two types, wet (ischemia with superimposed bacterial infection leading to liquefactive necrosis) and dry (ischemia leading to coagulative necrosis) [4]. Fat necrosis takes place in in acute pancreatitis. Fibrinoid necrosis is usually observed in blood vessels due to the deposition of immune complexes in blood vessel walls leading to leakage of fibrin.
2. What are Genes Involved in Necrosis?
As mentioned before, necrosis is always associated with an inflammatory response. Necrotic cells release factors such as HMGB1 and HDGF. These factors are sensed by a nod-like receptor protein 3 (NLRP3), which is a core protein of the inflammasome. Then, inflammasome is activated and the pro-inflammatory cytokine IL1βis released. NLRP3 inflammasome activation is primarily triggered by ATP produced by mitochondria released from damaged cells [5] [6].
Although, necrosis is caspase independent, and it appears in response to damage or pathology, not during normal development. Despite this, increasing evidence demonstrates that a programmed form of necrosis(called necroptosis) is very common in vivo, mainly in diverse forms of neurodegeneration and death inflicted by ischemia or infection. Unlike unordered necrosis, necroptosis is a more physiological and programmed type of necroptotic death and shares several key processes with apoptosis. CUSABIO lists partial popular targets related necrosis (including necroptosis), click to see all the related molecules/targets and research reagents of them.
References
[1] Ruffolo pr. the pathogenesis of necrosis. i. correlated light and electron microscopic observations of the myocardial necrosis induced by the intravenous injection of papain. am j pathol. 1964 nov; 45:741-56.
[2] Nabeeha Khalid; Mahzad Azimpouran. Necrosis [J]. Treasure Island (FL): StatPearls Publishing; 2022 Jan-.
[3] Sarhan M, Land WG, Tonnus W, Hugo CP, Linkermann A. Origin and Consequences of Necroinflammation [J]. Physiol Rev. 2018 Apr 01; 98 (2): 727-780.
[4] Taj-Aldeen SJ, Gene J, Al Bozom I, Buzina W, Cano JF, Guarro J. Gangrenous necrosis of the diabetic foot caused by Fusarium acutatum [J]. Med Mycol. 2006, Sep, 44 (6): 547-52.
[5] Nikoletopoulou V, Markaki M, Palikaras K, Tavernarakis N. Crosstalk between apoptosis, necrosis and autophagy [J]. Biochim Biophys Acta. 2013 Dec;1833 (12): 3448-3459.
[6] Fu C, Zhang X, Lu Y, Wang F, Xu Z, Liu S, Zheng H, Liu X. Geniposide inhibits NLRP3 inflammasome activation via autophagy in BV-2 microglial cells exposed to oxygen-glucose deprivation/reoxygenation [J]. Int Immunopharmacol. 2020 Jul; 84: 106547.