Pyrolysis
Pyroptosis (also known as inflammatory necrosis) was first proposed to describe the discovery of caspase-1-dependent cell death in macrophages by cookson’s research team in 2001 [1]. The morphological characteristics, occurrence and regulatory mechanism of pyroptosis are different from other types of cell death such as apoptosis and necrosis.
1. What are the Features of Pyrolysis?
Pyroptosis mainly mediates the activation of various caspases including Caspase-1 through the inflammasome, resulting in the splicing and multimerization of various Gasdermin family members including GSDMD. Then, cells perforate, causing cell death. Compared with apoptosis, pyroptosis occurs faster and is accompanied by the release of a large number of pro-inflammatory factors.
Morphologically, during cell pyroptosis, the cell swells, and before the cell ruptures, a protrusion is formed on the cell, and then pores are formed on the cell membrane, causing the cell membrane to lose its integrity, release its contents, and cause an inflammatory response. At this time, the nucleus is located in the center of the cell, and with the change of morphology, the nucleus shrinks and the DNA is broken.
2. What are the Main Pathways of Pyrolysis?
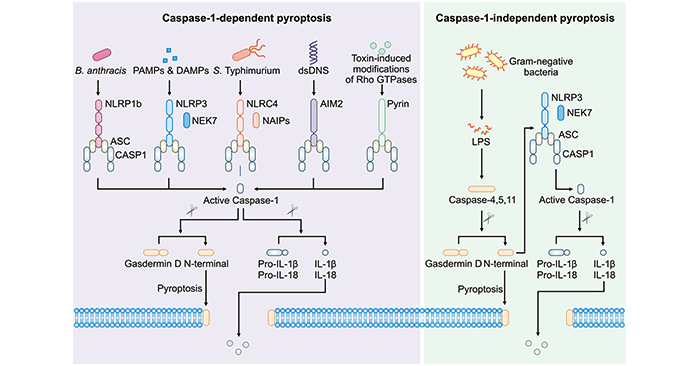
Pyroptosis is regulated via a caspase-1-dependent or caspase-1-independent pathway (As the figure 1 shows) [2]. Caspase-1-dependent pyroptosis, also known as canonical inflammasome pathway, requires activation of the canonical inflammasomes. In this pathway, pathogen-associated molecular patterns or danger-associated molecular patterns activate their pattern recognition receptors (PRRs) (including NLRP1b, NLRP3, NLRC4, AIM2 or Pyrin), respectively. Activation of the NLRP3 and NLRC4 inflammasomes requires the kinase NEK7 and ligand-binding NAIP proteins, respectively. Then, inflammasomes further recruit the inflammasome adaptor ASC and the cysteine protease caspase-1 to form macromolecular complex. Caspase-1 directly cleaves gasdermin D and the precursor cytokines pro-IL-1β and pro-IL-18, initiating pyroptosis and maturation of IL-1β and IL-18, respectively. The cleaved N-terminal portion of gasdermin D forms pores on the host cell membrane to mediate the release of cytoplasmic contents.
Caspase-1-independent pyroptosis, also known as non-canonical inflammasome pathway, is executed by human caspase-4, human caspase-5 or mouse caspase-11 [3]. These inflammatory caspases directly cleave gasdermin D and initiate pyroptosis. The N-terminal fragment also activates the NLRP3 inflammasome and caspase-1-dependent maturation of IL-1β and IL-18.
Figure 1. Diagram of caspase-1-dependent pyroptosis and caspase-1-independent pyroptosis
2. What are Genes Involved in Pyrolysis?
As mentioned on the last paragraph, the initiation and development of pyrolysis is regulated by multiple protein molecules or complexes, such as inflammasome, GSDMD, caspase-1, IL1β, etc.
Inflammasome is a multi-protein complex assembled by cytoplasmic pattern recognition receptors (PRRs), which include Toll-like receptors (TLRs), RIG-I-like receptors (RLRs) and NOD-like receptors (NLRs). Among of them, NLRP3 is a key effector of pyroptosis. It is an important part of the innate immune system and can recognize pathogens. Pathogen-associated molecular patterns (PAMPs) or host-derived damage-associated molecular patterns (DAMPs) recruit and activate the pro-inflammatory protease Caspase-1 to initiate signaling in the body's inflammatory response and antiviral immunity pathway that protects the host from infection.
GSDMD is a member of a conserved protein family, GSDM. In humans, GSDMD consists of a 242-amino acid amino-terminal domain and a 199-amino acid carboxy-terminal domain linked by 43 amino acids. The N-terminal domain is also called pore-forming domain (PFD) due to form GSDM pores in the plasma membrane. The C-terminal domain is also named repressor domain (RD) because of inhibiting the formation of perforations by the N-terminal domain. GSDMD is expressed in immune cells and intestinal epithelial cells. Cleavage of GSDMD is the mechanism of triggering pyroptosis by Caspase-1 and Caspase-11. GSDMD forms a pore as an effector of pyroptosis [4].
Caspase-1 usually exists in an inactive precursor form. After being activated by the inflammasome, Caspase-1 will cleave the sequence of GSDMD-linked amino acid to form a hydrophilic C-terminal domain and a lipophilic N-terminal domain. Moreover, IL-1β and IL-18 precursors are cleaved to mature forms by Caspase-1 and released through hollow annular pores formed by GSDM or during membrane rupture.
Caspase-11 can be directly activated by "invaders" such as bacteria, act independently on GSDMD and generate an N-terminal lipophilic domain, which oligomerizes and binds to the cell membrane to form a pyroptotic pore. In addition, Caspase-11 can directly recognize and bind cytosolic lipopolysaccharide (LPS), triggering IFN-γ to initiate pyroptosis.
CUSABIO lists partial popular targets related necrosis (including necroptosis), click to see all the related molecules/targets and research reagents of them.
References
[1] Cookson BT, Brennan MA. Pro-inflammatory programmed cell death [J]. Trenda Microbiol, 2001, 9: 113-114.
[2] Man SM, Karki R, Kanneganti TD. Molecular mechanisms and functions of pyroptosis, inflammatory caspases and inflammasomes in infectious diseases [J]. Immunol Rev. 2017;277(1):61-75.
[3] Stowe I, Lee B, Kayagaki N. Caspase-11: arming the guards against bacterial infection [J]. Immunological reviews. 2015, 265:75–84.
[4] Kovacs SB, Miao EA. Gasdermins: Effectors of Pyroptosis [J]. Trends Cell Biol. 2017, 27(9): 673-684.