Cell Adhesion Receptor
Cells are in constant contact with their surroundings, and these connections, mainly through cell adhesion, are critical for cell differentiation and growth. Cell adhesion includes intercellular adhesion and cell-matrix adhesion, the former refers to direct contact between cells, while the latter refers to the interaction between cells and the extracellular matrix. Cell adhesion connects cells in different ways (including tight junctions, adherent junctions, and gap junctions) and can participate in signal transduction for cells to detect and respond to changes in the surrounding environment.
Each type of cell adhesion is accomplished by specific membrane protein receptors, the adhesion receptors. Alterations in cell adhesion disrupt important cellular processes and contribute to a variety of diseases, including cancer and arthritis. Currently in the field of cancer, the more hot adhesion receptors are integrins and cadherins.
1. Integrin
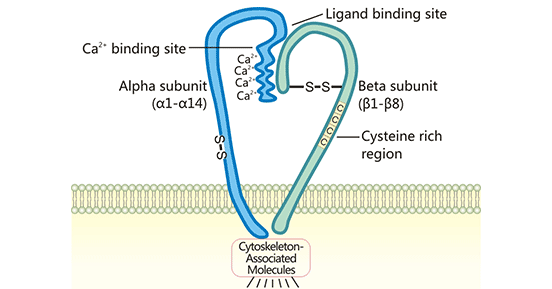
Integrin is the most typical adhesion receptor in cell-extracellular matrix adhesion, and its ligands are mainly ECM proteins (such as fibronectin, fibronectin, vitronectin, etc.) . Cell-to-extracellular matrix (EMC) adhesion refers to the binding of ECM proteins to adhesion receptors to generate bidirectional signals that regulate cell polarity, migration, survival, and proliferation. The members of the integrin family are all heterodimers composed of α and β polypeptide chains (or subunits). At least 14 α-chain subunits and 8 β-chain subunits are known in the integrin family. So far, more than 20 types of integrin family members have been found. According to the different β subunits, they can be divided into 8 groups from β1 to β8. The beta chains of the members of the same group are all the same, while the alpha chains are different. Most α-chain subunits can only bind to one β-chain subunit, while most β-subunits can bind to several different α-chain subunits. The αβ dimer of each integrin can bind multiple ECM proteins, and each ECM protein may also bind multiple α and β dimers. The effects of this ECM-integrin conjugate are dynamic and interconnected.
Figure 1. The typical structure of integrin
2. Cadherin
Cadherins, the canonical adhesion receptors that mediate intercellular adhesion, are a family of transmembrane receptors that form Ca2+-dependent homeoantigen dimers between two adjacent cells. The molecular structure of cadherin is highly homologous, and its extracellular part has 5 domains, 4 of which are homologous and contain Ca2+ binding sites. The site that determines the binding specificity of cadherin is in a domain near the N-terminal, and the binding specificity can be changed from E-cadherin to P-cadherin by changing only two amino acid residues. The cytoplasmic portion of the cadherin molecule is the most highly conserved region and is involved in signal transduction.
So far, more than 30 cadherins have been identified, which are distributed in different tissues. Among them, E-cadherin is the most important cadherin expressed in epithelial cells. E-cadherins dimers between adjacent cells inhibit tumor cell migration and proliferation through β-catenin and Hippo kinase-dependent processes
3. Popular Targets Related Cell Adhesion Receptor
CUSABIO has listed some popular targets related cell adhesion receptor. Click on the corresponding target to view all the reagents related to the target.