Enzyme-Linked Receptor
Enzyme-linked receptors, also known as catalytic receptors, are all one-time transmembrane proteins, and the intracellular domain often has a certain enzymatic activity. Enzyme-linked receptors have completely different molecular structures and properties from G protein-coupled receptors. This transmembrane signal transduction process does not require the participation of G proteins and the production of second messengers. The cytoplasmic side of the enzyme-linked receptor molecule has its own enzyme activity, or can directly bind and activate the enzyme in the cytoplasm, thereby realizing the regulation of cell function by extracellular signals.
Enzyme-linked receptors typically respond slowly to extracellular signaling molecules that regulate growth, reproduction, differentiation, and survival, and their functioning requires many steps of intracellular transduction, often resulting in changes in gene expression. But enzyme-linked receptors can also directly mediate the rapid deformation of the cytoskeleton, changing the way cells shape and move. Enzyme-linked receptors mainly include five types, namely receptor tyrosine kinase (RTK), receptor serine/threonine kinase (RSTK), receptor tyrosine phosphatase, receptor guanylate cyclase and tyrosine protein kinase associated receptors. Among them, the three types of enzyme-linked receptors involving the most drug targets are receptor tyrosine kinases, receptor serine/threonine kinases and receptor tyrosine phosphatases.
1. Receptor Tyrosine Kinase
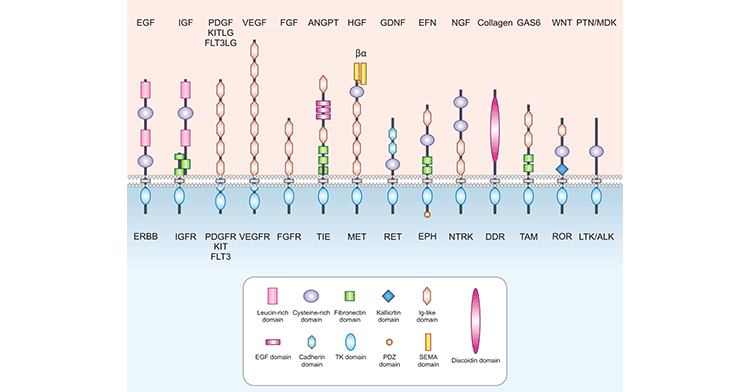
Receptor tyrosine kinases (RTKs), the largest class of enzyme-linked receptors, are receptors for growth factors and enzymes that catalyze the phosphorylation of downstream target proteins. At present, more than 50 RTKs have been found in vertebrates, which can be divided into 20 families, and there are more than 100 growth factors that can activate RTKs. All RTKs are composed of three parts: an extracellular domain, a single-pass transmembrane hydrophobic α-helical region, and an intracellular domain. The extracellular domain is the domain that binds ligands, containing the binding site for ligands, which are soluble, membrane-bound polypeptides or protein hormones, such as insulin and growth factors. The intracellular region contains tyrosine protein kinase activity, is the catalytic site of tyrosine protein kinase, and has an autophosphorylation site. Among them, the extracellular part of RTKs contains various domains with specific amino acid sequences (cysteine-rich domains, acidic domains, immunoglobulin-like domains, etc.). Figure 1 lists several common types of RTKs structures and corresponding receptors:
Figure 1. The diagram of RTKs structures
2. Receptor Serine/Threonine Kinases
Receptor serine/threonine kinase (RSTK) is a single-pass transmembrane protein receptor with serine/threonine protein kinase activity in the intracellular domain, which functions as a heterodimer. The main ligands are members of the TGFβs family, including TGF-β1-TGF-β5, which have similar structures and functions and have multiple effects on cells. Depending on the cell type, it may inhibit cell proliferation, stimulate extracellular matrix synthesis, stimulate bone formation, attract cells through chemotaxis, and act as an inducible signal during embryonic development.
3. Receptor Tyrosine Phosphatases
Receptor tyrosine phosphatase is a single-pass transmembrane protein receptor, and the intracellular region of the receptor has the activity of protein tyrosine phosphatase. The binding of extracellular ligands to the receptor stimulates the activity of the enzyme, making specific intracellular Dephosphorylation of phosphotyrosine residues in signaling proteins acts to control the lifespan of phosphotyrosine residues, resulting in lower levels of phosphotyrosine residues in quiescent cells. Its role is not simply the opposite of RPTK, but may work in concert with tyrosine kinases, such as in cell cycle regulation. CD45 on the surface of leukocytes belongs to this type of receptor, and the specific ligand is not known.
4. Popular Targets Related Enzyme-Linked Receptor
CUSABIO has listed some popular targets related enzyme-linked receptor. Click on the corresponding target to view all the reagents related to the target.