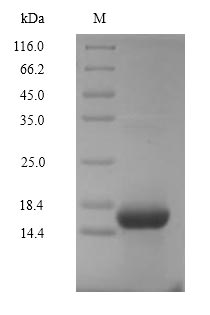
The production of the recombinant human complement C5 involves several steps. First, the gene fragment encoding the 678-751aa of C5 protein is isolated and co-inserted into a vector with the N-terminal 6xHis-Myc-tag gene. The vector acts as a carrier that delivers the targeted gene into mammalian cells for protein expression. The recombinant human C5 protein is extracted from the cell lysate and then purified through affinity chromatography. Its purity is over 90% as measured by SDS-PAGE.
Human Complement C5 is a vital component of the complement system, comprising two polypeptide chains, C5 alpha and C5 beta. C5 alpha can be further processed into C5 alpha' and C5a. It plays a significant role in complement-mediated diseases and immune responses [1][2][3]. Eculizumab, a humanized monoclonal antibody targeting C5, has been effectively used to manage complement-mediated diseases such as paroxysmal nocturnal hemoglobinuria (PNH) and atypical hemolytic uremic syndrome (aHUS) by inhibiting the cleavage of C5 into C5a and C5b [4][5][6]. Inhibition of C5 cleavage is a pivotal strategy in averting complement-mediated endothelial injury and thrombotic microangiopathy [7][8].
References:
[1] R. DiScipio, L. Jenner, S. Thirup, L. Sottrup-Jensen, J. Nyborg, & E. Stura, Crystallization of human complement component c5, Acta Crystallographica Section D Biological Crystallography, vol. 54, no. 4, p. 643-646, 1998. https://doi.org/10.1107/s0907444997015011
[2] T. Contractor, S. Kobayashi, E. Silva, R. Clausen, C. Chan, E. Vosburghet al., Sexual dimorphism of liver metastasis by murine pancreatic neuroendocrine tumors is affected by expression of complement c5, Oncotarget, vol. 7, no. 21, p. 30585-30596, 2016. https://doi.org/10.18632/oncotarget.8874
[3] N. Cooper and H. Müller‐Eberhard, The reaction mechanism of human c5 in immune hemolysis, The Journal of Experimental Medicine, vol. 132, no. 4, p. 775-793, 1970. https://doi.org/10.1084/jem.132.4.775
[4] L. Zhang, W. Qiao, S. Crooke, Y. Li, A. Abid, B. Xuet al., Development of autologous c5 vaccine nanoparticles to reduce intravascular hemolysis in vivo, Acs Chemical Biology, vol. 12, no. 2, p. 539-547, 2017. https://doi.org/10.1021/acschembio.6b00994
[5] S. Nandavaram, Thrombotic microangiopathy in solid organ transplantation, Obm Transplantation, vol. 08, no. 02, p. 1-37, 2024. https://doi.org/10.21926/obm.transplant.2402211
[6] C. Mache, B. Acham-Roschitz, V. Frémeaux‐Bacchi, M. Kirschfink, P. Zipfel, S. Roedlet al., Complement inhibitor eculizumab in atypical hemolytic uremic syndrome, Clinical Journal of the American Society of Nephrology, vol. 4, no. 8, p. 1312-1316, 2009. https://doi.org/10.2215/cjn.01090209
[7] A. Jin, L. Boroujerdi-Rad, G. Shah, & J. Chen, Thrombotic microangiopathy and human immunodeficiency virus in the era of eculizumab, Clinical Kidney Journal, vol. 9, no. 4, p. 576-579, 2016. https://doi.org/10.1093/ckj/sfw035
[8] L. Catanese, K. Link, & H. Rupprecht, Microangiopathy in multiple myeloma: a case of carfilzomib-induced secondary thrombotic microangiopathy successfully treated with plasma exchange and complement inhibition, BMC Nephrology, vol. 24, no. 1, 2023. https://doi.org/10.1186/s12882-023-03228-9