Norovirus (NoV) is a non-enveloped, single-stranded positive-sense
RNA virus and one of the leading pathogens of acute gastroenteritis worldwide. It causes approximately 685
million infections annually, making it the primary cause of non-bacterial gastroenteritis outbreaks and a
significant contributor to sporadic gastroenteritis in both children and adults. The virus is primarily
transmitted via the fecal-oral route, including through contaminated food (such as lettuce and seaweed),
water, or person-to-person contact.
1. Virus Structure
Norovirus belongs to the family Caliciviridae, a small, non-enveloped RNA virus with
a diameter of approximately 26–34 nm. The viral particles exhibit a typical cup-shaped structure, with a
capsid featuring T=3 icosahedral symmetry, assembled from 180 identical VP1 proteins.
The structure of a virus primarily consists of:
VP1: Major Capsid Protein;
VP2: Minor Capsid Protein;
Non-structural proteins (NS1/2–NS7): Involved in viral replication and assembly.
The VP1 protein can be further divided into two main structural domains:
Capsid domain: Forms the inner, tightly packed spherical framework of the capsid, which
encloses and protects the viral genome.
Protruding domain: Located outside the shell domain, it forms an outward-extending
arched protrusion and is a key region for virus-host cell interactions. The protruding domain can be further
subdivided into two subdomains, P1 and P2, with the P2 subdomain situated at the outermost part. It serves
as the primary site determining viral antigen diversity, recognizing host tissue blood group antigens, and
binding neutralizing antibodies [1].
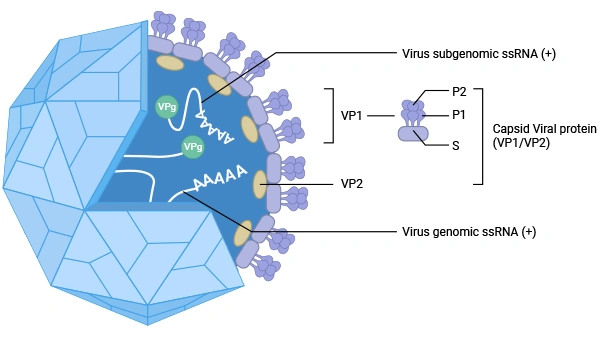
Figure 1. The Norovirus (NoV) virion
Based on sequence similarity analysis of VP1 and NS7 polymerase, noroviruses can be
classified into ten distinct genogroups (GI–GX), which can be further subdivided into 49 genotypes. Among
these, only the GI, GII, and GIV genogroups cause acute gastroenteritis in humans. The GII. 4 genotype is
associated with severe clinical courses and has given rise to six pandemic strains, while GII. 17 is another
genotype recently detected with high transmissibility.
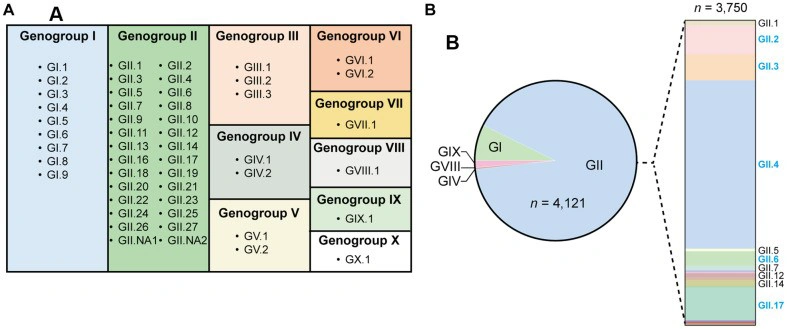
Figure 2. Schematic overview of norovirus classification showing the ten
recognized genogroups (GI–GX) and their respective genotypes.
Image from: PMID: 41218799
CUSABIO Recombinant proteins related to norovirus research
2. Norovirus Pathogenesis
The pathogenesis of norovirus is multifactorial.The outcomes of collaboration can be
primarily categorized into the following levels:
2.1 Viral level: NS1 inhibits interferon, NS3 induces cell lysis, and capsid
mutations enhance virulence.
- NS1/2, upon cleavage by caspase-3, releases the NS1 protein, which subsequently
inhibits the intestinal interferon-λ (IFN-λ) response in its secreted form, thereby aiding the virus in
evading immune clearance.
- The N-terminal domain of NS3 mimics the host MLKL protein, targeting the
mitochondrial membrane lipid cardiolipin, leading to mitochondrial dysfunction and cell lysis, promoting
viral release, and inducing cell death.
- The capsid protein VP1 activates PKD2 via the TLR2/MyD88/IRAK4 pathway, inhibits the
nuclear translocation of the transcription factor AP2γ, downregulates the expression of AQP3 in intestinal
epithelial cells, reduces water reabsorption, and triggers diarrhea.
2.2 Host level: HBGA/FUT2 genetic background, immune deficiency status,
age-related susceptibility (e. g., passive absorption in young hosts).
- Norovirus primarily infects tuft cells in the intestinal tract, which express the
viral receptors CD300lf (for murine norovirus) or histo-blood group antigens (HBGAs, for human norovirus)
[2].
- Cluster cells constitute an extremely low proportion in the intestine (<1%), but
viruses can persistently infect and colonize them over the long term, leading to chronic viral shedding.
Human norovirus also relies on HBGA as a binding receptor. Individuals with a deficiency in the FUT2 gene,
which encodes α(1, 2) fucosyltransferase, are naturally resistant to infection due to the lack of HBGA
expression [3].
- Type I Interferons (IFN-I): Protect the host from severe enteritis, but Type III
Interferons (IFN-λ) may exacerbate diarrhea symptoms.
- CD8+ T cells: Mediate epithelial damage in chronic infections, leading to villous
atrophy (as seen in CVID-associated enteropathy).

Figure 3. Presumed pathogenesis of CVID‐associated enteropathy complicated by
chronic norovirus infection.
Image from:PMID: 41229388
2.3 Environmental aspects: microbial regulation, aerosol transmission, and
shellfish enrichment.
- Intestinal microbiota stimulate tuft cell proliferation through type 2 cytokines
(such as IL-4/IL-13), indirectly enhancing viral replication.
- Certain probiotics (such as Limosilactobacillus fermentum PV22) can inhibit viral
activity by secreting γ-aminobutyric acid [4].
- Norovirus is highly stable in the environment: it can withstand a pH 2.7 acidic
environment for 3 hours, heating at 60°C for 30 minutes, survive in frozen foods for 6 months, and persist
on contaminated surfaces for ≥7 days.
- An extremely low infectious dose (18–2, 800 viral particles) is sufficient to cause
illness, and transmission can occur via aerosols (vomitus), contaminated water/food, and contact.
3. Transmission route
Norovirus is one of the primary pathogens of acute gastroenteritis worldwide, with
an extremely high transmission capacity, capable of triggering large-scale outbreaks in various
environments. Understanding its complex transmission pathways is crucial for developing effective prevention
and control strategies.
Main transmission routes
Contact transmission
Contact transmission is the most basic and common way for norovirus to spread between people,
primarily including direct and indirect contact.
Direct contact: Having close contact with symptomatic or asymptomatic infected individuals.
Indirect contact: Contact with virus-contaminated surfaces (such as door handles, tabletops,
toys, etc.), followed by infection through the hand-to-mouth route.
Foodborne Transmission
Consuming food or water contaminated with norovirus is the primary cause of large-scale outbreaks.
Sources of Pollution: Viruses can be introduced into the food chain by infected food
handlers during food preparation or through contaminated irrigation water, shellfish, and other
sources.
Waterborne Transmission
Drinking or coming into contact with water sources contaminated with feces (such as well water or
recreational water) can lead to norovirus outbreaks. Additionally, the virus may persist throughout
the entire process of sewage collection, treatment, and discharge, causing ongoing contamination of
the environment and water sources through various pathways.
Aerosol/Airborne Transmission
When patients vomit or have diarrhea, they can produce aerosols containing viral particles. Studies
using feline calicivirus (FCV) as a surrogate for norovirus have confirmed the possibility of aerosol
transmission and the effectiveness of corresponding air purification technologies.
4. Virus Detection Technology
Molecular Biology Testing: Real-time fluorescent quantitative PCR
technology, with its high sensitivity and specificity, is the gold standard for clinical diagnosis of
norovirus. By continuously optimizing detection methods and primer design, the accuracy and efficiency of
detection can be improved.
Immunological Testing: Such as enzyme-linked immunosorbent assay (ELISA) and
colloidal gold immunochromatography, which are simple to operate and suitable for batch screening. By
developing antibodies with high sensitivity and specificity, the accuracy and reliability of immunological
detection can be improved.
New Detection Technologies: For instance, metagenomic sequencing technology
enables the direct detection of norovirus in clinical samples without the need for culturing, providing
robust support for epidemic surveillance and source tracing.
FAQ
Q:Why do some people not get infected even after exposure to norovirus?
This is related to an individual's genetic background, particularly the functional status of the
FUT2 gene. The FUT2 gene encodes α(1, 2) fucosyltransferase, which is responsible for
synthesizing histo-blood group antigens (HBGAs) on the surface of intestinal cells. HBGAs serve as key
receptors for norovirus recognition and infection of host cells. If an individual carries an
inactivating mutation in the FUT2 gene (i. e., a "non-secretor" individual), they are unable to
express HBGAs on the surface of intestinal epithelial cells. As a result, the virus cannot effectively
bind to and invade these cells, granting these individuals natural resistance to certain norovirus
strains, such as Norwalk virus.
Q:Why can norovirus survive for so long in the environment?
Due to its lack of an envelope structure and its strong environmental tolerance. Norovirus
lacks a lipid envelope, making it highly resistant to environmental factors such as dryness, acids,
alkalis, and temperature. It can survive for 3 hours in an acidic environment with a pH of 2.7, remain
infectious after heating at 60°C for 30 minutes, persist in frozen foods for up to 6 months, and
survive on contaminated surfaces for more than 7 days. This stability makes it easily transmissible
through food, water, and contact, and extremely difficult to eradicate completely.
Q:Why does diarrhea occur after norovirus infection?
Diarrhea is a direct result of viruses interfering with the absorption of water in the
intestines. The VP1 capsid protein of norovirus can activate PKD2 through the TLR2/MyD88/IRAK4
signaling pathway, thereby inhibiting the nuclear translocation of the transcription factor AP2γ,
leading to downregulation of aquaporin AQP3 expression in intestinal epithelial cells. AQP3 is
responsible for water reabsorption, and its reduced expression decreases the intestinal water
absorption capacity, ultimately causing water retention in the intestinal lumen and triggering
diarrhea.
Q:Why Does Norovirus Repeatedly Infect the Same Person?
Because the virus has multiple genotypes and the immune protection period is short. Norovirus
can be classified into multiple genogroups such as GI, GII, and GIV, each of which includes various
genotypes (e. g., GII. 4, GII. 17, etc.). There are significant antigenic differences among different
genotypes, meaning that antibodies produced after infection with one genotype may be ineffective
against another. Additionally, the immune protection period for norovirus typically lasts only 6
months to 2 years, allowing individuals to experience multiple infections throughout their lifetime.
References:
[1] Winder N, Gohar S, Muthana M. Norovirus: An Overview of Virology and
Preventative Measures. Viruses 2022; 14(12): 2811. doi:10.3390/v14122811
[2] Kennedy EA, et al. Age-associated features of norovirus infection analysed in mice. Nature Microbiol
2023; 8: 1023–1032. doi:10.1038/s41564-023-01383-1
[3] Lindesmith L, et al. Human susceptibility and resistance to Norwalk virus infection. Nature Med 2003;
9: 548–553. doi:10.1038/nm860
[4] Prasad BV, et al. Norovirus replication, host interactions and vaccine advances. Nat Rev Microbiol
2025; 23: 456–472. doi:10.1038/s41579-024-01144-9




