Lassa fever is an acute viral hemorrhagic fever caused by the Lassa
virus (LASV), primarily prevalent in West Africa, including Nigeria, Sierra Leone, Liberia, and Guinea. The
virus exists in at least seven distinct lineages. (For example:I-VII), each lineage exhibits a regional
distribution.,Related to rodent population migration。
1. Lassa Virus Structure
Lassa virus is a highly pathogenic arenavirus to humans.。The viral particles are
generally spherical or pleomorphic, with a diameter of approximately 80-150 nanometers. They are enveloped
by a lipid bilayer derived from the host cell's plasma membrane. Embedded in the envelope are multiple
glycoprotein spikes (GPC), which are the core structures responsible for recognizing host cell receptors and
mediating membrane fusion for cellular entry. Beneath the envelope lies a matrix layer formed by the Z
protein. Inside, the viral RNA genome, nucleoprotein (NP), and RNA-dependent RNA polymerase (L protein)
together constitute the ribonucleoprotein complex.
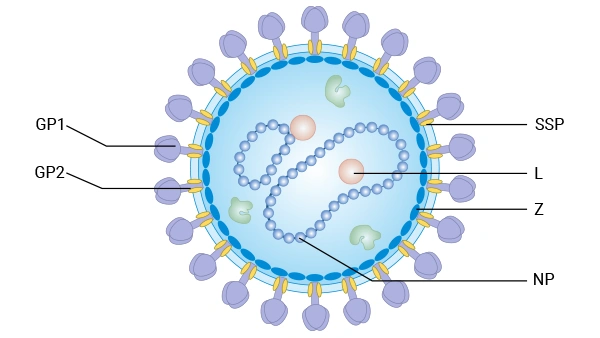
Image caption: Schematic representation of arenavirus virions.
CUSABIO Lassa Recombinant proteins related to virus research
| Target |
Code |
Product name |
Source |
Tag Info |
| GPC |
CSB-BP322925LCP1
|
Recombinant Lassa virus Pre-glycoprotein polyprotein GP complex (GPC), partial |
Baculovirus |
N-terminal 10xHis-tagged and C-terminal Myc-tagged |
| GPC |
CSB-CF322925LCP
|
Recombinant Lassa virus Pre-glycoprotein polyprotein GP complex (GPC), partial |
in vitro E.coli expression system |
N-terminal 10xHis-tagged |
| N |
CSB-EP318401LNP
|
Recombinant Lassa virus Nucleoprotein (N) |
E.coli |
N-terminal 6xHis-SUMO-tagged |
| N |
CSB-EP318401LNPe1
|
Recombinant Lassa virus Nucleoprotein (N) |
E.coli |
Tag-Free |
| GPC |
CSB-EP322925LCP1
|
Recombinant Lassa virus Pre-glycoprotein polyprotein GP complex (GPC),
partial |
E.coli |
N-terminal 10xHis-tagged and C-terminal Myc-tagged |
| GPC |
CSB-EP362480LNP2
|
Recombinant Lassa virus Pre-glycoprotein polyprotein GP complex (GPC),
partial |
E.coli |
N-terminal 10xHis-tagged and C-terminal Myc-tagged |
| N |
CSB-YP318401LNP
|
Recombinant Lassa virus Nucleoprotein (N) |
Yeast |
N-terminal 6xHis-tagged |
| N |
CSB-BP356406LCP
|
Recombinant Lassa virus Nucleoprotein (N) |
Baculovirus |
N-terminal 10xHis-tagged and C-terminal Myc-tagged |
| Z |
CSB-EP524825LNP
|
Recombinant Lassa virus RING finger protein Z (Z) |
E.coli |
N-terminal 6xHis-tagged |
| GPC |
CSB-CF362480LNP
|
Recombinant Lassa virus Pre-glycoprotein polyprotein GP complex (GPC)
|
in vitro E.coli expression system |
N-terminal 10xHis-tagged |
2. Transmission routes
It is primarily transmitted through contact with the excretions of virus-carrying
rodents, particularly the Mastomys natalensis. Human-to-human transmission can also occur through direct
contact with the bodily fluids of infected individuals.Or mother-to-child
transmissionOccurred[1]. Additionally,Research has found that other potential rodent species,
such as the Mastomys erythroleucus and the Lophuromys sikapusi, may also carry LASV, particularly in regions
like Sierra Leone and Liberia. The expansion of these species' habitats, such as through forest degradation,
could accelerate the spread of the virus to new areas [2]. It causes 100, 000 to 300, 000
infections annually, resulting in approximately 5, 000 deaths. In hospitals within endemic areas, 11% of
fever cases can be confirmed as Lassa fever.
3. Clinical Manifestations and Diagnosis
Symptoms:Typical manifestations include fever, fatigue, headache, nausea, and
vomiting. Severe cases may present with bleeding tendencies, neurological symptoms, and multiple organ
failure.Early stage of the diseaseLassa fever and other infectious diseases, such as malaria, typhoid fever,
shigellosis, yellow fever, and other viral hemorrhagic fevers.The symptoms are very similar and difficult to
distinguish clinically.Approximately 32% of pediatric patients have detectable viral RNA in their
cerebrospinal fluid, with viral loads typically higher than in plasma, indicating direct infection of the
central nervous system.
The following diagnostic methods can be employed to confirm whether the symptoms are
caused by Lassa virus infection:
| Detection Type |
Detection target |
Applicable Scenarios |
Advantage |
Limitations |
| Nucleic Acid Test (RT-PCR) |
Viral RNA |
Early diagnosis of acute infection, quantification of viral load, epidemiological surveillance.
|
High sensitivity, strong specificity, quantifiable, short window period |
Requires professional equipment and personnel, high sample processing standards, and relatively
high
costs. |
| Antigen Detection (ELISA/Rapid Test) |
Viral proteins (NP, GPC, etc.) |
Rapid screening for acute infections, on-site testing in resource-limited areas. |
Easy to operate, fast detection, low cost, and no need for complex equipment. |
Lower sensitivity than nucleic acid testing and a longer window period compared to nucleic acid
testing.
|
| Antibody Detection (ELISA) |
IgM/IgG |
Diagnosis during the recovery period, retrospective investigation of past infections,
seroepidemiological survey |
Distinguishes recent infection (IgM) from past infection (IgG), easy to operate. |
Early infection has a window period and cannot distinguish between current infection and past
infection.
|
| Virus isolation and culture |
Live virus |
Scientific Research, Virus Strain Identification, Vaccine and Drug Development Verification |
Obtain live virus strains, gold standard method |
Requires BSL-4 laboratory, time-consuming, high operational risk, not suitable for routine
clinical
diagnosis. |
4. Pathogenesis
Imbalance in immune response: Severely ill patients exhibit expansion of
myeloid-derived suppressor cells, alterations in the secondary lymphoid organ stromal network, and
antigen-specific T cell unresponsiveness, which are similar to the immunosuppressive mechanisms observed in
sepsis and cancer [3].
Virus-host interactions: The spike complex on the virus surface mediates cell
entry by recognizing matriglycan (a linear carbohydrate) on the host cell surface [4]. The signal
peptide plays a crucial role in stabilizing the native conformation of the spike complex.
5. Key Characteristics Comparison Between Lassa Virus and Other Hemorrhagic Fever
Viruses
| Features |
Lassa virus |
Ebola virus |
Marburg virus |
Yellow fever virus |
| Virus family |
Arenaviridae |
Filoviridae |
Filoviridae |
Flaviviridae |
| Main epidemic areas |
West Africa |
Central Africa, West Africa |
Central Africa, East Africa |
Tropical Africa, Central America, South America |
| Natural Host |
Rodents (Multimammate MiceMastomys natalensisetc.) |
Fruit bat (PteropodidaeSection) |
Fruit bat (Rousettus aegyptiacus) |
Primates, mosquitoes (humans as accidental hosts) |
| Transmission routes |
Contact infection through rodent excreta; human-to-human transmission
via bodily fluids. |
Direct contact with the blood, secretions, or organs of an infected
person |
Direct contact with the blood or secretions of an infected person |
Aedes mosquito bite (Aedes aegypti); Non-direct interpersonal
communication |
| Basic Reproduction Number (R₀) |
1.0–1.6 |
1.5–2.5 |
1.4–2.0 |
3.0–6.0 |
| Average case fatality rate |
Approximately 1% |
25–90% |
23–90% |
20–50% |
| Incubation period |
6–21 days |
2–21 days |
2–21 days |
3–6 days |
| Typical Clinical Manifestations |
Fever, sore throat, nausea and vomiting, bleeding tendency (relatively
rare), neurological symptoms |
Sudden high fever, severe bleeding, multiple organ failure, shock |
Sudden high fever, severe bleeding, multiple organ failure, shock |
Fever, jaundice, bleeding, and impairment of liver and kidney function
(the term "yellow fever" is derived from the symptom of jaundice). |
| Laboratory Diagnosis |
RT-PCR, ELISA (antigen/antibody), virus isolation |
RT-PCR, ELISA (antigen/antibody), virus isolation |
RT-PCR, ELISA (antigen/antibody), virus isolation |
RT-PCR, ELISA (IgM/IgG), virus isolation |
| Treatment methods |
Supportive therapy; Ribavirin (efficacy uncertain) |
Supportive care; monoclonal antibodies (such as REGN-EB3), remdesivir
(limited data) |
Supportive care; no approved specific antiviral drugs are currently
available. |
Supportive care; no specific antiviral drugs. |
| Vaccine |
No approved vaccines; |
Approved vaccine (rVSV-ZEBOV, Ervebo®) |
No approved vaccines; |
Effective vaccine (17D attenuated live vaccine,YF-VAX®) |
| Biosafety Level |
BSL-4 |
BSL-4 |
BSL-4 |
BSL-3 |
6. Treatment
There is currently no licensed vaccine for Lassa fever, but several potential
candidates are under development. Early intensive supportive care with fluid management and symptomatic
treatment improves survival rates.
The antiviral drug ribavirin has been used to treat Lassa fever; however, there is
considerable uncertainty regarding its efficacy in Lassa fever patients and the optimal dosing regimen.
Other candidate treatment options are at various stages of development and evaluation.
FAQ
Q:What are the main structural proteins of the Lassa virus?
Lassa virus primarily consists of four core structural proteins: GPC, NP, RNA-dependent RNA
polymerase (L protein), and matrix protein (Z). GPC is responsible for recognizing host cell
receptors and mediating membrane fusion for viral entry into cells; NP binds to viral RNA to form
ribonucleoprotein complexes; the L protein is involved in the transcription and replication of the
viral genome; the Z protein participates in viral assembly and budding processes.
Q:How does Lassa virus enter host cells?
The virus mediates cellular entry by recognizing matriglycan (a linear carbohydrate) on the host
cell surface through its surface glycoprotein spike (GPC). After binding to the host receptor,
GPC triggers the membrane fusion process, releasing the viral ribonucleoprotein complex into the
cytoplasm and initiating infection.
Q:Why is Lassa fever difficult to diagnose early?
The early symptoms of Lassa fever are highly similar to those of other tropical infectious
diseases, making clinical differentiation difficult. Typical symptoms such as fever, fatigue,
and headache overlap with diseases like malaria, typhoid fever, and yellow fever, making it necessary
to rely on laboratory tests such as RT-PCR, ELISA, or virus isolation for definitive diagnosis.
Q:Why does Lassa fever lead to severe illness?
Severely ill patients primarily exhibit an imbalance in immune responses, particularly the
suppression of specific T-cell functions. In severe infections, the expansion of myeloid-derived
suppressor cells leads to anergy in virus-specific T cells, preventing the body from effectively
clearing the virus and subsequently triggering severe complications such as multi-organ failure.
Q:Does Lassa virus have neuroinvasive potential?
Lassa virus can directly invade the central nervous system, particularly in pediatric
patients. Clinical studies indicate that viral RNA can be detected in the cerebrospinal fluid of
approximately 32% of pediatric patients, with viral loads often higher than those in plasma,
suggesting the virus has the ability to directly infect the central nervous system.
References:
[1]Garry RF, et al. Lassa fever - the road ahead. Nature Reviews Microbiology
2023; 21(2): 87–96. doi:10.1038/s41579-022-00789-8.
[2] Adesina AS, et al. Circulation of Lassa virus across the endemic Edo-Ondo axis, Nigeria, with
cross-species transmission between multimammate mice. Emerging Microbes & Infections 2023; 12(1): 2219350.
doi:10.1080/22221751.2023.2219350.
[3] Lafoux B, et al. Expansion of myeloid suppressor cells and suppression of Lassa virus-specific T
cells during fatal Lassa fever. PLoS Pathogens 2025; 21(4): e1013111. doi:10.1371/journal.ppat.1013111.
[4] Katz M, et al. Structure and receptor recognition by the Lassa virus spike complex. Nature 2022;
603(7902): 565–570. doi:10.1038/s41586-022-04429-2.


