Recently, the WHO has reported an outbreak of hantavirus infection
on a polar expedition cruise ship that departed from Argentina on April 1. Starting from April 6, the virus
has infected eight people, and as of May 7, three deaths have been recorded, raising public concern.
1. What is Hantavirus?
Hantaviruses belong to the Hantaviridae family within the order Bunyavirales. The
virion structure of these viruses exhibits unique complexity, consisting of a lipid envelope, surface
glycoprotein spikes, and an internal tripartite negative-sense RNA genome. These components work in concert
to drive the entire viral life cycle, including host recognition, membrane fusion, and genome replication.
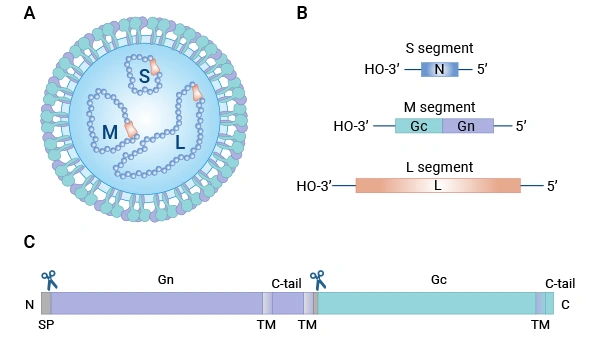
Figure 1. Schematic representation of hantavirus particle and genes.
Image source:PMID: 31439149
Research has found that there are over 20 types of hantaviruses that can cause
distinct diseases, with significant differences in clinical manifestations depending on the virus species.
They are primarily classified into hemorrhagic fever with renal syndrome and hantavirus pulmonary syndrome
[1].
| Disease Type |
Hemorrhagic fever with renal syndrome (HFRS) |
Hantavirus Pulmonary Syndrome (HPS) |
| Main endemic regions |
China, South Korea, Russia, Europe, etc. |
The Americas (e. g., Argentina, the United States, etc.) |
| Major virus types |
Hantaan virus, Seoul virus, etc. |
Andes virus, Sin Nombre virus, etc. |
| Target organ |
Kidney |
Lung |
| Main clinical features |
"Three Pains" (headache, lower back pain, orbital pain) + "Three Reds" (flushing of the face,
neck, and chest) + bleeding + kidney damage。 |
Fever, dry cough → respiratory distress, pulmonary edema, shock |
| Typical disease course |
Febrile phase → Hypotensive shock phase → Oliguric phase → Polyuric phase → Convalescent phase
|
Sudden onset → rapid respiratory failure |
| Case fatality rate |
Approximately 0.1% - 1% |
Gundam 30% - 40% |
2. Pathogenesis and Immune Response
Pathogenesis
Hantavirus (represented by pathogenic types such as HTNV and ANDV) infection the
core pathological event is Loss of vascular endothelial barrier integrity, leading to plasma extravasation,
hemorrhage, and shock. The virus itself does not directly lyse endothelial cells, but rather through
Immune-mediated injury Causeing dysfunction.
The virus enters the human body through inhalation of aerosols contaminated by the
excreta of infected rodents, initially infecting alveolar macrophages and dendritic cells. Subsequently, the
virus utilizes receptors such as β3 integrin (αVβ3) and DEC-205/DC-SIGN to enter vascular endothelial cells,
macrophages, and renal tubular epithelial cells for efficient replication.
It is worth noting that the virus exhibits a "latent infection" characteristic in
endothelial cells without directly causing cytopathic effects. After endothelial cells are infected with
hantavirus, they secrete vascular endothelial growth factor (VEGF), which triggers the downregulation of
VE-cadherin in adherens junctions, thereby disrupting tight junctions between endothelial cells and leading
to increased vascular permeability. At the same time, infected endothelial cells also produce
pro-inflammatory cytokines and chemokines, such as IP-10 and RANTES, and upregulate cell surface adhesion
molecules. As the viral load increases, the host's immune system becomes overactivated. Recruited monocytes
and macrophages polarize into a pro-inflammatory M1 phenotype, releasing large amounts of inflammatory
cytokines such as TNF-α, IL-6, and CXCL10, thus forming a "cytokine storm." In addition, CD8+ T cells cause
secondary damage to the endothelial barrier during the clearance of virus-infected endothelial cells,
ultimately leading to severe clinical consequences such as significantly increased vascular permeability,
plasma extravasation, shock, kidney injury, and hemorrhage [2].
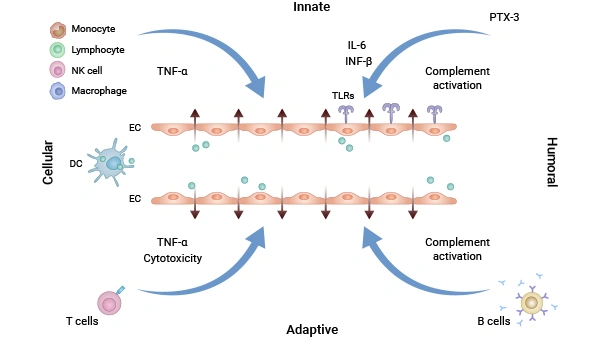
Figure 2. Both humoral and cellular components of the innate and adaptive immune
systems can contribute to vascular leakage. In response to hantavirus-infected endothelial cells (ECs),
neutrophils release inflammatory cytokines such as TNF-α, which directly or indirectly enhance vascular
permeability.
Image source:PMID 35384417
This pathological process is not driven by direct viral lysis of cells but rather
results from a dysregulated host immune response, which also provides a critical intervention point for
targeted immunomodulation—such as restoring interferon signaling, inhibiting RIPK3, or mimicking the natural
host's inflammatory self-limiting mechanisms.
Immune Response - Cytokine
Storm
For more details, please click: What You Have to Know about Cytokine Storm and Virus
Infection
In hantavirus infection, this immune system "storm" is not driven by a single factor
but results from the combined action of multiple key molecules and signaling pathways, exacerbating fatal
outcomes such as vascular leakage and multi-organ failure. The essence of a cytokine storm is the excessive
production of pro-inflammatory factors. The core cytokines involved in the viral immune response fall into
the following main categories:
- Interleukin (IL) family: In severe and fatal cases of HFRS, researchers have
identified specific changes in the IL profile. Specifically, IL-18 levels significantly increased, while
IL-7 levels decreased. Another key molecule is IL-33, which plays the role of a "cytokine storm initiator"
in hantavirus infection [3].
- Tumor Necrosis Factor (TNF): Activated neutrophils release TNF-α, which can
directly or indirectly enhance vascular permeability by mediating other inflammatory factors, and this is
a key step leading to hemorrhage and shock.
- Chemokines and growth factors: Levels of CCL-5 (also known as RANTES) and stem
cell growth factor (SCGF)-β were also significantly elevated in fatal cases.
- Cytokines are not generated out of thin air; their explosive synthesis depends on
the activation of a series of signaling pathways within the cell. These pathways include the NF-κB pathway, the
interferon (IFN) signaling pathway, the STING pathway, and VEGF signaling pathway.
Intervening in these pathways is key to controlling cytokine storms. The structural proteins GP and NP of
hantavirus are core targets for vaccine development and research on immune mechanisms.
CUSABIO provides the following professional tools to support research in viral immunology and related therapies:
● Hantavirus ELISA kits
● Hantavirus recombinant protein
Targets in Cytokine Storm
3. Transmission Routes of Hantavirus
The main transmission routes of hantavirus involve rodents and include the following
three modes:
Respiratory transmission
Inhalation of aerosols generated from urine, feces, or saliva of infected rodents.
Digestive tract transmission
Ingestion of food or water contaminated with excreta from infected rodents.
Contact transmission
Being bitten or scratched by infected rodents, or direct/indirect contact of broken skin or mucous
membranes with rodent excreta or contaminated materials.
Hantavirus infection generally does not spread from person to person, and routine social
interactions or public contact do not pose a risk of transmission.
4. Epidemiology Geographical Distribution
Hantavirus infections exhibit a wide geographical distribution globally, with an
estimated 200,000 infection cases worldwide each year. Its epidemiological characteristics are influenced by
various factors, including climate, environment, social development, the ecology of rodent hosts, and human
behavior [4]. In the Americas, Sin Nombre virus (SNV) is the primary pathogen causing hantavirus
cardiopulmonary syndrome (HCPS). As of January 2020, 143 laboratory-confirmed cases have been identified in
Canada alone.
In China, the main hosts include the striped field mouse (Apodemus agrarius) and the
brown rat (Rattus norvegicus). A spatiotemporal predictive analysis of data from China between 2005 and 2028
indicates that viruses carried by the brown rat (Rattus) will be the most active in terms of transmission in
the future, with risks even surpassing those in regions dominated by the striped field mouse (Apodemus)
[5]. In Europe, such as in Poland, hantavirus infections are primarily caused by the Puumala and
Dobrava serotypes. In Russia, over 130,000 cases of HFRS have been reported in the past 20 years, with 98.4%
occurring in western Russia. Differences in serotypes have led to significant variations in mortality rates.
5. Biomarkers and Clinical Diagnosis
The clinical manifestations of HFRS are diverse, typically including fever, myalgia,
headache, lower back pain, and gastrointestinal symptoms.Since viremia appears in the early stages of the
disease and can persist for more than two weeks, viral load has become an important indicator for early
diagnosis. Studies have found that viral load is positively correlated with white blood cell, lymphocyte,
and monocyte counts, but negatively correlated with platelet count [6].
In addition to directly detecting the virus,Thrombomodulin (TM), Plasma BNP level,
Procalcitonin (PCT), Osteoprotegerin (OPG) Multiple biomarkers have been confirmed.It has significant
predictive value for assessing the severity of the disease. In addition, leukocytosis, thrombocytopenia,
proteinuria, hematuria, coagulation abnormalities, and electrolyte disturbances (such as hyponatremia and
hypocalcemia) are also common laboratory features in patients with HFRS, including children. For adult
patients, hypotensive shock, myoglobin levels, and neutrophil counts have also been identified as
independent factors for predicting the risk of critical illness. Currently, there is still a lack of
specific etiological therapy for hantavirus infection. Animal model studies have provided directions for
treatment: a targetedHantavirusThe neutralizing monoclonal antibody HCO2 against the virus, when
administered on the fifth day post-infection, can also alleviate weight loss and renal medullary hemorrhage
in mice, though earlier administration yields better results. This suggests that the timing of immunotherapy
is crucial.
CUSABIO focuses on key nodes in the immunopathology of hantavirus infection. We are
committed to simulating the natural host's inflammatory self-limiting mechanism to block the "cytokine
storm" at its root, providing full-chain support for Hemorrhagic Fever with Renal Syndrome (HFRS) and
Hantavirus Pulmonary Syndrome (HPS), from biomarker discovery to early development of antibody drugs.
FAQ: HantavirusVirus Frequently Asked Questions
Q:Why does hantavirus infection cause severe vascular leakage and even shock?
Vascular leakage caused by hantavirus infection is primarily due to uncontrolled host immune
responses rather than direct viral destruction of cells. The virus does not directly lyse
endothelial cells; instead, it activates the immune system after infection, inducing a cytokine storm,
recruiting inflammatory cells, and disrupting the connections between vascular endothelial cells,
ultimately leading to plasma extravasation, decreased blood pressure, and shock.
Q:Why do the types and severity of diseases caused by different hantaviruses vary significantly?
Different virus types exhibit varying preferences for target organs, leading to two distinct
clinical manifestations: hemorrhagic fever with renal syndrome (HFRS) and hantavirus pulmonary
syndrome (HPS). There are over 20 pathogenic types of hantavirus. For instance, the Hantaan
virus primarily attacks the kidneys, causing HFRS, while the Andes virus mainly targets the lungs,
resulting in HPS. The two differ significantly in disease progression, symptom characteristics, and
case fatality rates (approximately 0.1–1% for HFRS vs. 30–40% for HPS).
Q:Is there a specific therapeutic drug for hantavirus at present?
Currently, there is no specific antiviral therapy, and immunomodulation along with early
neutralizing antibody intervention are important treatment directions. Clinical management
remains primarily supportive; however, in animal models, administration of neutralizing monoclonal
antibodies (such as HCO2) during the early stages of infection can reduce renal hemorrhage and weight
loss, suggesting that targeting immune responses or antibody therapy holds potential value.
Q:How to accurately diagnose hantavirus infection at an early stage?
Early diagnosis primarily relies on viral load testing, combined with the assessment of multiple
blood and urine biomarkers to evaluate disease severity. In the early stages of onset, viremia
can persist for more than two weeks. Viral load is positively correlated with white blood cell and
lymphocyte counts and negatively correlated with platelet count. Meanwhile, markers such as
thrombomodulin, procalcitonin, and osteoprotegerin can be used to assess the risk of disease severity.
Q:What key tools can CUSABIO provide for hantavirus research?
CUSABIO provides a comprehensive range of research tools, including ELISA detection kits for key
cytokines such as TNF-α, IL-6, IL-18, and RANTES; recombinant proteins for hantavirus GP and N
proteins (suitable for vaccine and immune mechanism research); and a full series of anti-payload
antibodies required for ADC drug development.
References:
[1] Tkachenko E, et al. Clinical Manifestations of Hemorrhagic Fever with
Renal Syndrome, Various Nosologic Forms and Issues of Hantavirus Infections Terminology. Viruses.
2025;17(4):578. doi:10.3390/v17040578
[2] Su L, et al. Dynamic alterations of circulating lymphocytes during the trajectory of Hantaan
virus-induced hemorrhagic fever with renal syndrome. Front Immunol. 2025;16:1354892.
doi:10.3389/fimmu.2025.1354892
[3] Garanina E, et al. Cytokine Storm Combined with Humoral Immune Response Defect in Fatal
Hemorrhagic Fever with Renal Syndrome Case, Tatarstan, Russia. Viruses. 2019;11(7):601.
doi:10.3390/v11070601
[4] Vial PA, et al. Hantavirus in humans: a review of clinical aspects and management. Lancet Infect
Dis. 2023;23(9):e371-e382. doi:10.1016/S1473-3099(23)00128-7
[5] Wang Y, et al. Spatiotemporal trends of hemorrhagic fever with renal syndrome (HFRS) in China
under climate variation. Proc Natl Acad Sci USA. 2024;121(4):e2312556121. doi:10.1073/pnas.2312556121
[6] Ma S, et al. Temporal Dynamics of Viremia in Hemorrhagic Fever With Renal Syndrome Patients. J
Med Virol. 2026;98(5):e70963. doi:10.1002/jmv.70963