Endometrial Cancer
Endometrial cancer is the most common gynecologic malignancy in the United States. This cancer begins in the uterus, which is the hollow, pear-shaped pelvic organ where fetal development occurs. The incidence of endometrial cancer among malignant gynecological tumors has increased with lifestyle and environmental changes, and has reached almost half of all cases of uterine cancer in Western countries [1].
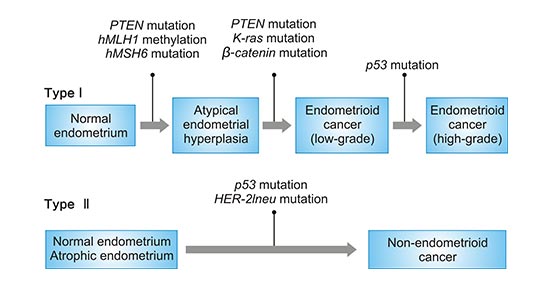
Traditional classification of endometrial cancer is usually based on clinical and endocrine features: types I and II. Type I endometrial carcinomas generally develop before the time of menopause. Type II endometrial carcinomas occur in older, post-menopausal women in most cases. Regarding to the mechanisms of carcinogenesis in endometrial cancer, genetic abnormalities (mutations in the DNA) of cells in the endometrium are currently clear. The figure 1 shows that several gene mutations have emerged as candidates for roles in carcinogenesis of type I and II endometrial cancer, based on observation of the mutation in endometrial hyperplasia and at least a similar incidence of mutation in endometrial cancer.
Figure 1. Gene mutations in the carcinogenesis of endometrial cancer
*this diagram is derived from publication on The Journal of Obstetrics and Gynaecology Research [2]
In this article, we list part of these proteins involved in endometrial cancer based on the information provided by NCG (web resource to analyze duplicability, orthology and network properties of cancer genes).
Here, we display several key targets involved in mechanism of endometrial cancer, including:
-
PPP2R1A (Protein Phosphatase 2 Scaffold Subunit Aalpha) is a subunit of protein phosphatase 2, a complex implicated in the negative control of cell growth and division. PPP2R1A mutations are common in the serous type of endometrial cancer. Moreover, the PPP2R1A mutation regions are conserved among species and known to interact with the regulatory subunits of the PP2A enzyme [3]. Furthermore, Dorien Haesen et al. revealed that recurrent PPP2R1A mutations in uterine cancer act through a dominant-negative mechanism to promote malignant cell growth [4].
-
ARID1A (AT-rich interactive domain-containing protein 1A) is a subunit of several different SWI/SNF protein complexes, which play a key role in regulating gene activity (expression) by a process known as chromatin remodeling. Mutation of ARID1A induces changes in expression of multiple genes (CDKN1A, SMAD3, MLH1 and PIK3IP1) via chromatin remodeling dysfunction and contributes to carcinogenesis. It has been shown to cause transformation of cells in association with the PI3K/AKT pathway [5]. Recently, ARID1A loss has been found in every type of endometrial cancer, indicating its important role in initiation and progression of this disease [6].
-
CHD4 (chromodomain helicase DNA binding protein 4) mediates proliferation and migration of non-small cell lung cancer via the RhoA/ROCK pathway by regulating PHF5A. CHD4, a core subunit of the nucleosome remodeling and deacetylation (NuRD) complex, is frequently mutated in these patients with endometrial cancer. And Yang Li's study indicated that mutations in CHD4 can promote endometrial tumorigenesis by increasing CSC character through TGFβ signaling pathway [7].
-
FBXW7 (F-box/WD repeat-containing protein 7) is a component of the ubiquitin ligase complex, which functions in phosphorylation-dependent ubiquitination. Diego A. Garcia-Dios et al. found that mutations in PIK3CA, TP53, PTEN and FBXW7 correlate with high tumor grade, endometrial cancer type and lymph node status, whereas PIK3CA H1047R mutations serve as prognostic markers for relapse-free survival in endometrial cancer patients [8].
-
SPOP (Speckle Type BTB/POZ Protein) may modulate the transcriptional repression activities of death-associated protein 6 (DAXX). It is an adapter protein of the CUL3-based E3 ubiquitin ligase complexes. It selectively recruits substrates for their ubiquitination and subsequent degradation. Recently, several exome-sequencing studies of endometrial cancer revealed high frequency somatic mutations in SPOP (5.7-10%) [9].
References
[1] Kouji Banno, Megumi Yanokura, Yusuke Kobayashi et al. Endometrial Cancer as a Familial Tumor: Pathology and Molecular Carcinogenesis (Review) [J]. Curr Genomics. 2009, 10(2): 127–132.
[2] Kouji Banno, Megumi Yanokura, Miho Iida et al. Carcinogenic mechanisms of endometrial cancer: Involvement of genetics and epigenetics [J]. The Journal of Obstetrics and Gynaecology Research. 2014.
[3] Haesen D, Abbasi Asbagh L, Derua R et al. Recurrent PPP2R1A Mutations in Uterine Cancer Act through a Dominant-Negative Mechanism to Promote Malignant Cell Growth [J]. Cancer Res. 2016, 76(19): 5719-5731.
[4] Nagendra DC, Burke J 3rd, Maxwell GL, Risinger JI. PPP2R1A mutations are common in the serous type of endometrial cancer [J]. Mol Carcinog. 2012, 51(10):826-31.
[5] TAKASHI TAKEDA, KOUJI BANNO, RYUICHIRO OKAWA et al. ARID1A gene mutation in ovarian and endometrial cancers (Review) [J]. Oncol Rep. 2016, 35(2): 607–613.
[6] CHRISAVGI TOUMPEKI, ANASTASIOS LIBERIS, IOANNIS TSIRKAS et al. The Role of ARID1A in Endometrial Cancer and the Molecular Pathways Associated With Pathogenesis and Cancer Progression [J]. In Vivo. 2019, 33(3): 659–667.
[7] Yang Li, Qingxin Liu, Daniel J McGrail et al. CHD4 mutations promote endometrial cancer stemness by activating TGF-beta signaling [J]. Am J Cancer Res. 2018, 8(5): 903- 914.
[8] Garcia-Dios, Diego A.; Lambrechts, Diether; Coenegrachts, Lieve et al. High-throughput interrogation of PIK3CA, PTEN, KRAS, FBXW7 and TP53 mutations in primary endometrial carcinoma [J]. Gynecologic Oncology. 2013, 128(2), 327–334.
[9] P Zhang, K Gao, X Jin et al. Endometrial cancer-associated mutants of SPOP are defective in regulating estrogen receptor-α protein turnover [J]. Cell Death Dis. 2015, 12;6(3):e1687.