Colorectal Cancer
Colon cancer is a type of cancer that begins in the large intestine (colon). The colon is the final part of the digestive tract. Colorectal cancer is the third most common cancer in the Western hemisphere and the incidence increases with increasing age [1]. The environmental and genetic factors that cause colorectal cancer do so by promoting the acquisition of hallmark behaviors of cancer in colon epithelial cells. Many studies have observed the loss of genomic and/or epigenomic stability in the majority of early neoplastic lesions in the colon (namely, aberrant crypt foci, adenomas and serrated polyps), which is likely a central molecular and pathophysiological event in the initiation and formation of colorectal cancer [2] [3].
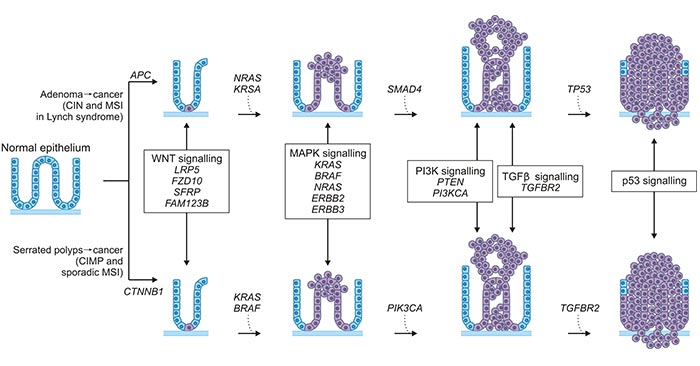
Figure 1. The polyp to colorectal cancer sequences
*this diagram is derived from publication on Adv Cancer Res [4]
Currently, two discrete normal colon to colorectal cancer sequences have been identified (Figure 1). Both sequences involve the progression of normal colon epithelial cells to aberrant crypt foci, followed by early and advanced polyps with subsequent progression to early cancer and then advanced cancer. The 'classic' or traditional pathway (top) involves the development of tubular adenomas that can progress to adenocarcinomas. An alternate pathway (bottom) involves serrated polyps and their progression to serrated colorectal cancer has been described in the last 5–10 years. Figure 1 indicates the genes mutated or epigenetically altered of each sequence. As you see, some genes are shared between the two pathways (such as KRAS) whereas others are unique (such as BRAF).
In this article, we list part of targets involved in colorectal cancer based on the information provided by NCG (web resource to analyze duplicability, orthology and network properties of cancer genes).
Colorectal cancer progression is driven by a series of well-defined genetic alterations, including mutations in APC, BRAF, KRAS, PIK3CA, p53 and FBXW7. Here, we display several key targets involved in mechanism of colorectal cancer, including:
-
APC (adenomatous polyposis coli) function has been linked to the Wnt signal transduction pathway. In normal condition, APC can lead to degradation of α-catenin and to cellular adhesion, migration and apoptosis [5] [6] [7]. Since APC inactivation is sufficient to initiate tumor development, APC mutation affect degradation of α-catenin and truncated APC fragments stimulate colorectal cancer cell migration and generate chromosomal instability [8].
-
KRAS (Kirsten rat sarcoma viral oncogene homolog), a member of MAP kinase (MAPK) pathway, is a small GTPase involved in intracellular signal transduction [9]. KRAS mutations contribute to the deregulation of KRAS protein activity, which results in the loss of GTPase activity and gain of oncogenic activity. Point mutations in exons 2 and 3 of the KRAS gene are responsible for malignant transformation. KRAS mutations are observed in approximately 37.4% of the Iranian patients with colorectal cancer [10].
Moreover, KRAS mutations in colorectal cancer have generally been found to be inversely correlated with BRAF mutations [11]. Additionally, Li et al. previously demonstrated that KRAS/PIK3CA bi-mutations are significantly more frequent in patients with stage IV colorectal cancer, compared to lower-stage carcinomas. KRAS/PIK3CA bi-mutations may induce metastasization through a synergic effect in the activation of PI3K-AKT pathway [12] [13].
-
SMAD4 (SMAD Family Member 4), also called mothers against decapentaplegic homolog 4, is a highly conserved protein present in all metazoans. It belongs to the SMAD family of transcription factor proteins, which act as mediators of TGF-β signal transduction. Reports of loss of SMAD4 protein expression in colorectal cancer range from 9% to 66% (9.3%, 14%, 66%), and it is a common feature of early-onset colorectal tumors as it is in colorectal cancers diagnosed in other age-groups [14].
-
FBXW7 (F-box and WD repeat domain-containing 7), a member of F-box family proteins, constitute one subunit of Skp1, Cul1, and F-boxprotein (SCF) ubiquitin ligase complex. It is a potential tumor suppressor that regulates ubiquitination and proteolysis of multiple targets such as cyclin E, c-Myc, c-Jun and Notch [15].
References
[1] Sigurdis Haraldsdottir, Hulda M Einarsdottir, Agnes Smaradottir et al. Colorectal cancer – review [J]. Laeknabladid. 2014, 100(2):75-82.
[2] Colussi D, Brandi G, Bazzoli F et al. Molecular pathways involved in colorectal cancer: implications for disease behavior and prevention [J]. Int J Mol Sci. 2013, 14(8):16365-85.
[3] Grady WM, Carethers JM. Genomic and epigenetic instability in colorectal cancer pathogenesis [J]. Gastroenterology. 2008, 135(4):1079-99.
[4] Ernst J. Kuipers, William M. Grady, David Lieberman et al. Colorectal cancer [J]. Nat Rev Dis Primers. 2015, 1: 15065.
[5] Yang J, Zhang W, Evans PM et al. Adenomatous polyposis coli (APC) differentially regulates beta-catenin phosphorylation and ubiquitination in colon cancer cells [J]. J Biol Chem. 2006, 30; 281(26):17751-7.
[6] Etienne-Manneville S, Hall A. Cdc42 regulates GSK-3beta and adenomatous polyposis coli to control cell polarity [J]. Nature. 2003, 421(6924):753-6.
[7] Watanabe T, Wang S, Noritake J et al. Interaction with IQGAP1 links APC to Rac1, Cdc42, and actin filaments during cell polarization and migration [J]. Dev Cell. 2004, 7(6):871-83.
[8] M Toma, D Cimponeriu, A Pompilia et al. Molecular analysis of mutations for the adenomatous polyposis coli (APC) gene in Romanian patients with colorectal cancer [J]. J Med Life. 2008, 1(4): 423–428.
[9] Velho S, Moutinho C, Cirnes L et al. BRAF, KRAS and PIK3CA mutations in colorectal serrated polyps and cancer: Primary or secondary genetic events in colorectal carcinogenesis [J]? BMC Cancer. 2008, 8:255.
[10] Atena Irani Shemirani, Mahdi Montazer Haghighi, Saman Milanizadeh et al. The role of kras mutations and MSI status in diagnosis of colorectal cancer [J]. Gastroenterol Hepatol Bed Bench. 2011, 4(2): 70–75.
[11] Daniele Calistri, Claudia Rengucci, Ian Seymour et al. KRAS, p53 and BRAF gene mutations and aneuploidy in sporadic colorectal cancer progression [J]. Cellular Oncology. 2006, 28: 161–166.
[12] Li HT, Lu YY, An YX et al. KRAS, BRAF and PIK3CA mutations in human colorectal cancer: relationship with metastatic colorectal cancer [J]. Oncol Rep. 2011, 25(6):1691-7.
[13] Luca Reggiani Bonetti, Valeria Barresi, Antonino Maiorana et al. Clinical Impact and Prognostic Role of KRAS/BRAF/PIK3CA Mutations in Stage I Colorectal Cancer [J]. Dis Markers. 2018, 2018: 2959801.
[14] S. G. Royce, K. Alsop, A. Haydon et al. The role of SMAD4 in early-onset colorectal cancer [J]. Colorectal Dis. 2010, 12(3): 213–219.
[15] Hao Liu, Kai Wang, Haixiao Fu et al. Low expression of the ubiquitin ligase FBXW7 correlates with poor prognosis of patients with colorectal cancer [J]. Int J Clin Exp Pathol. 2018, 11(1): 413–419.