Lung Cancer
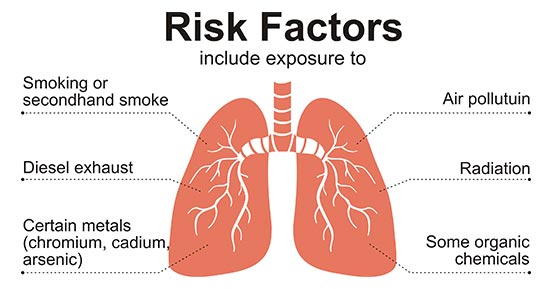
Lung cancer is a type of cancer that starts in the lungs. It causes cells to divide in the lungs uncontrollably and form tumors to reduce a person's ability to breathe. Worldwide, about three quarters of lung cancers are attributable to smoking; others are caused by occupational workplace exposure, radon exposure, and air pollution (Figure 1). It is more common in men, and incidence increases with age [1] [2].
Figure 1. the risk factors of getting lung cancer
Lung cancer is a heterogeneous disease comprising several subtypes with pathologic and clinical relevance. Small-cell lung carcinoma (SCLC, 15% of all lung cancer) and non-small-cell lung carcinoma (NSCLC, 85%) are the two major forms of lung cancer. Non-small-cell lung cancer (NSCLC) is further classified into squamous-cell carcinoma, adenocarcinoma, and large-cell carcinoma. NSCLC, as the most frequent subtypes, has become important as a determinant of therapy in this disease. In recent years, the identification of molecular abnormalities in a large proportion of patients with lung cancer has allowed the emergence of personalized targeted therapies. In this article, we list part of targets involved in lung cancer based on the information provided by NCG (web resource to analyze duplicability, orthology and network properties of cancer genes).
Here, we display several key targets involved in mechanism of lung cancer, including:
-
TP53, also called p53, is a stress response protein that mediates the transcription of genes in response to genotoxic stress, oncogenic signaling, DNA damage, and cellular injury. Almost all small-cell lung cancers and more than half of NSCLCs harbor alterations in TP53. Apart from the loss of tumor-suppressor functions, TP53 mutations may result in gain of function favoring cellular proliferation, inhibition of apoptosis, and genomic instability [3]. Currently, Eprenetapopt, a tumor protein p53 stimulants, has been entered the phase III in clinic developed by Aprea Therapeutics for lung cancer treatment.
-
KRAS (Kirsten rat sarcoma viral oncogene homolog) is a member of the canonical RAS family that also includes HRAS and NRAS. Three decades ago, the importance of RAS in cancer pathogenesis was first recognized in rats. Now, RAS mutations have been found in approximately 30% of all human cancers, with KRAS as the most commonly mutated family member [4]. Cancer-causing mutations in RAS drastically impair the GTPase activity, resulting in RAS proteins that are locked in the active GTP-bound conformation, regardless of the upstream signal. Studies undertaken over the past decades have produced a wealth of information regarding the clinical relevance of KRAS mutations in lung cancer. Currently, several KRAS inhibitors development have entered the clinical stages, including AMG-510, MRTX-849, D-1553, LY-3499446, AZD-4785, Antroquinonol, GDC-6036 and JAB-3312. Among of them, the development of AMG-510 and MRTX-849 has entered phase III in clinic.
-
EGFR (Epidermal growth factor receptor) is a transmembrane protein with cytoplasmic kinase activity that transduces important growth factor signaling from the extracellular environment to the cell. Several studies have showed that more than 60% of NSCLCs express EGFR. And now, EGFR has become an important therapeutic target for the treatment of these tumors [5]. At present, many antagonists that target the kinase domain of EGFR have been developed and are approved for lung cancer treatment, such as Almonertinib Mesilate, Dacomitinib, Brigatinib, Olmutinib, Necitumumab, and so on.
-
KEAP1 (Kelch-like ECH-associated protein 1), a newly identified Nrf2-associated protein, functions as a sensor for electrophilic/oxidative stresses. Electrophilic/oxidative stresses are sensed by Keap1, which activates Nrf2 to achieve cytoprotection by regulating the expression of drug-metabolizing and antioxidative stress enzymes/proteins. Because oxidative and electrophilic stresses cause many diseases, including cancer, an abnormality in the Nrf2-KEAP1 system is hypothesized to facilitate the growth of cancer cells [5]. And accumulating evidence has demonstrated that KEAP1 is the third most frequently mutated gene in LUAD [6].
-
RB1 (Retinoblastoma transcriptional corepressor 1) is a RB pocket protein that plays an important role in cell cycle progression by interacting with various transcriptional factors as well as a tumor suppressor [7]. Small cell lung cancer (SCLC) accounts for 15% of lung cancers and is almost always linked to inactivating RB1 and TP53 mutations [8].
References
[1] Malhotra J, Malvezzi M, Negri E et al. Risk factors for lung cancer worldwide [J]. Eur Respir J. 2016, 48:889-902.
[2] Richard D, Fei Sun, Jon D et al. Lung cancer [J]. BMJ. 2019, 365: 1725.
[3] Ramaswamy Govindan and Jason Weber. TP53 Mutations and Lung Cancer: Not All Mutations Are Created Equal [J]. Clin Cancer Res. 2014, 20(17):4419–21.
[4] Peter M. K. Westcott and Minh D. To. The genetics and biology of KRAS in lung cancer [J]. Chin J Cancer. 2013, 32(2): 63–70.
[5] Tsutomu Ohta, Kumiko Iijima, Mamiko Miyamoto et al. Loss of Keap1 Function Activates Nrf2 and Provides Advantages for Lung Cancer Cell Growth [J]. Cancer Res. 2008. 68(5):1303–9.
[6] Rodrigo Romero, Volkan I. Sayin, Shawn M. Davidson et al. Keap1 loss promotes Kras-driven lung cancer and results in a dependence on glutaminolysis [J]. Nat Med. 2017. 23(11): 1362–1368.
[7] Priyanka Bhateja, Michelle Chiu , Gary Wildey et al. Retinoblastoma mutation predicts poor outcomes in advanced non small cell lung cancer [J]. Cancer Med. 2019. 8(4):1459-1466.
[8] Matthew G Oser, Raquel Fonseca, Abhishek A Chakraborty et al. Cells Lacking the RB1 Tumor Suppressor Gene Are Hyperdependent on Aurora B Kinase for Survival [J]. Cancer Discov. 2019. 9(2):230-247.