Breast Cancer
Breast cancer is the most frequent malignancy in women worldwide and is curable in ~70–80% of patients with early-stage, non-metastatic disease. More than 1.3 million women worldwide are diagnosed with breast cancer each year, making it the second most common form of cancer behind lung cancer [1] [2].
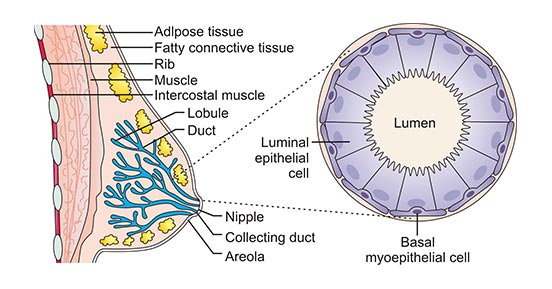
All breast cancers arise in the terminal duct lobular units (the functional unit of the breast) of the collecting duct (Figure 1). Currently, clinical practice typically uses a surrogate classification of five subtypes on the basis of histological and molecular characteristics, including triple-negative, HER2-enriched (non-luminal), luminal B-like HER2+, luminal B-like HER2– and luminal A-like breast cancer.
Figure 1. The structure of breast
*this diagram is derived from publication on Nature Reviews [3]
The surrogate intrinsic subtypes are typically used clinically and are based on histology and immunohistochemistry expression of key proteins, including oestrogen receptor (ER), progesterone receptor (PR), human epidermal growth factor receptor 2 (HER2) and the proliferation marker Ki67. Tumors expressing ER and/or PR are termed ‘hormone receptor-positive’; tumors not expressing ER, PR and HER2 are called ‘triple-negative’.
In addition to the three key proteins mentioned previous, there are still many proteins that play an important role in the development of breast cancer. In this article, we list part of these proteins involved in breast cancer based on the information provided by NCG (web resource to analyze duplicability, orthology and network properties of cancer genes).
Here, we display several key targets involved in mechanism of breast cancer, including:
-
ERBB2 (receptor tyrosine-protein kinase), also known as CD340 or HER2, is a protein in humans encoded by the ERBB2 gene. In normal cells, HER2 plays an important role in different cellular processes and maintaining the expression level of HER2 protein. However, overexpression of the HER2 gene or protein can lead to tumor formation and migration. After HER2 forms a dimer, it inhibits autophagy to promote cell migration.
Accumulating evidence shows that 25%~30% of breast cancer patients are HER2 positive. Moreover, currently the most targeted drugs for breast cancer have been developed with ERBB2 as the target, involving Trastuzumab, Pertuzumab, Ado-trastuzumab emtansine, Inetetamab and Trastuzumab biosimilar.
-
PIK3CA (phosphatidylinositol-4,5-bisphosphate 3-kinase, catalytic subunit alpha) gene provides instructions for making the p110 alpha (p110α) protein, which is one piece (subunit) of an enzyme called phosphatidylinositol 3-kinase (PI3K). PIK3CA is one of the most frequently mutated genes in invasive breast cancer (IBC), and is a biomarker of poor prognosis. These mutations are generally associated with hyper-activation of the phosphatidylinositol 3-kinase signaling pathway, which involves increased phosphorylation of AKT (p-AKT) [4]. Currently, Alpelisib, a phosphatidylinositol 3 kinase alpha inhibitor, has been approved for breast cancer treatment. Furthermore, there are three phosphatidylinositol 3 kinase alpha inhibitors in the phase III of clinic for breast cancer treatment, including Taselisib, Dactolisib and Buparlisib.
-
TP53 (Tumor protein P53), also known as p53, acts as a tumor suppressor, which means that it regulates cell division by keeping cells from growing and dividing (proliferating) too fast or in an uncontrolled way. TP53 mutations are the most frequent genetic alterations in invasive breast cancer. Although mutated in 30% of breast carcinomas, p53 is mutated in approximately 80% of triple-negative (TN) tumors [5]. Currently, there is a P53 gene stimulant (named INGN 225) that has entered the development of phase II in clinic for breast cancer treatment.
-
AKT1 (RAC-alpha serine/threonine-protein kinase), one of the three isoforms in the Akt family, encodes a protein called kinase B alpha (PKBα). Activated AKT1 phosphorylates a large number of downstream substrates that play a crucial role in regulating cell growth, metabolism, proliferation, apoptosis, angiogenesis, and drug responses [6]. Many studies have demonstrated that AKT1 is a suppressor of tumor metastasis in breast cancer [7]. Akt1 activation inhibits cell motility and invasion in breast cancer cells. Currently, Ipatasertib Dihydrochloride and Capivasertib, as AKT1 inhibitors, have entered the development of phase III in clinic.
-
GATA3 is a member of the GATA family of six zinc finger–containing transcription factors found to be essential in the development of luminal breast epithelium. It is highly expressed by the luminal epithelial cells in the breast [8]. The highest levels of GATA3 have been observed in the luminal types of breast cancers, and its expression decreases with increasing tumor grade [9]. Furthermore, high GATA3 expression was significantly associated with improved survival outcomes in premenopausal women with ER-positive breast cancer but not in postmenopausal patients [10].
References
[1] Grayson M. Breast cancer [J]. Nature. 2012, 485(7400): S49.
[2] Woolston C. Breast cancer [J]. Nature. 2015, 19;527(7578):S101.
[3] Nadia Harbeck, Frédérique Penault-Llorca, Javier Cortes, et al. Breast cancer [J]. Nature review. 2019, 5:66.
[4] Marie Colombe Agahozo, Anieta M. Sieuwerts, S. Charlane Doebar et al. PIK3CA mutations in ductal carcinoma in situ and adjacent invasive breast cancer [J]. Endocr Relat Cancer. 2019, 26(5):471-482.
[5] Michael J Duffy, Naoise C Synnott and John Crown. Mutant p53 in breast cancer: potential as a therapeutic target and biomarker [J]. Breast Cancer Res Treat. 2018, 170(2):213-219.
[6] Andrés López-Cortés , Paola E. Leone , Byron Freire-Paspuel, et al. Mutational Analysis of Oncogenic AKT1 Gene Associated with Breast Cancer Risk in the High Altitude Ecuadorian Mestizo Population [J]. BioMed Research International. 2018.
[7] Wei Li, Jiu-Zhou Hou, Jie Niu et al. Akt1 inhibition promotes breast cancer metastasis through EGFR-mediated β-catenin nuclear accumulation [J]. Cell Communication and Signaling. 2018, 16(82).
[8] Ashley Cimino-Mathews, Andrea P Subhawong, Peter B Illei et al. GATA3 expression in breast carcinoma: utility in triple-negative, sarcomatoid, and metastatic carcinomas [J]. Hum Pathol. 2013, 44 (7): 1341-9.
[9] Yoon NK Maresh EL Shen D et al. Higher levels of GATA3 predict better survival in women with breast cancer [J]. Hum Pathol. 2010, 41:1794–1801.
[10] Hosoda M Yamamoto M Nakano K et al. Differential expression of progesterone receptor, FOXA1, GATA3, and p53 between pre- and postmenopausal women with estrogen receptor-positive breast cancer [J]. Breast Cancer Res Treat. 2014, 144: 249–261.