MET, also known as hepatocyte growth factor receptor, serves as a critical receptor tyrosine kinase that orchestrates cellular responses to HGF signaling. This proto-oncogene plays fundamental roles in cell proliferation, survival, and motility, making it a focal point in cancer research where aberrant MET activation drives tumor progression, metastasis, and therapeutic resistance across multiple malignancies.
This recombinant monoclonal antibody, clone 2D12, offers the reproducibility that demanding research requires. Generated through recombinant technology with a defined sequence, it eliminates the lot-to-lot variability inherent in traditional hybridoma production, ensuring your experimental conditions remain consistent across long-term studies and collaborative projects.
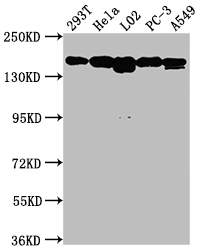
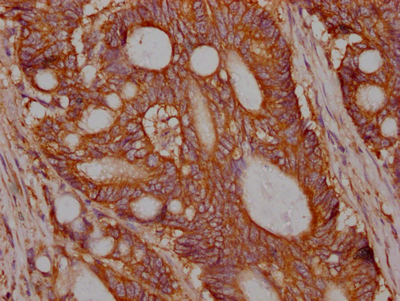
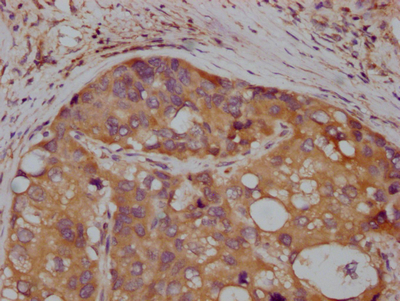
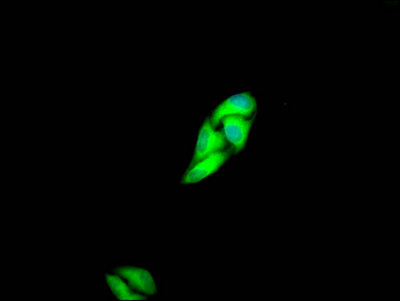
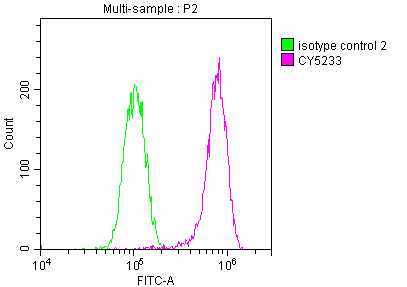
Validated across multiple platforms, this antibody demonstrates genuine versatility in your workflow. Western blot analysis confirms robust detection of MET at the expected 156 kDa band across diverse human cell lines including 293T, HeLa, L02, PC-3, and A549, with effective dilutions ranging from 1:500 to 1:5000. The observation of a single predominant band at 156 kDa, despite multiple predicted sizes, reflects the mature processed form most commonly detected under standard conditions. Immunohistochemistry validation in human colon cancer and liver cancer tissues demonstrates clear membrane and cytoplasmic staining patterns consistent with MET's known localization. Additional validation through immunofluorescence in HeLa cells reveals subcellular distribution patterns, while flow cytometry enables quantitative analysis of MET expression at the single-cell level.
For researchers investigating receptor tyrosine kinase signaling, oncogenic transformation, or therapeutic target validation, this antibody provides a reliable tool for examining MET biology across expression analysis, tissue characterization, and cell-based assays in human samples.